Tetramethylgermanium
 | |
| Names | |
|---|---|
| IUPAC name
Tetramethylgermane[1]
| |
| Other names | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.011.587 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Ge(CH3)4 |
| Molar mass | 132.770 g·mol−1 |
| Appearance | Colorless oily liquid[3][4][5] |
| Odor | Sweetish, chloroform-like[6] |
| Density | 0.978 g/cm3[7] |
| Melting point | −88 °C (−126 °F; 185 K)[7] |
| Boiling point | 43.4 °C (110.1 °F) at 987 hPa[8] |
| Solubility | Soluble in ethanol, diethyl ether[6] |
| Vapor pressure |
|
Refractive index (nD)
|
1.389 at 20 °C (68 °F)[5][8] |
| Structure | |
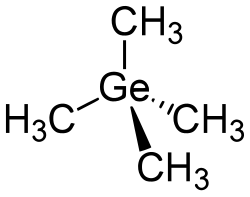
Molecular shape
|
Tetrahedral at Ge and C |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Toxic, flammable, can cause serious eye damage |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Danger |
Hazard statements
|
H225, H302, H312, H319, H332 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P261, P264, P264+P265, P270, P271, P280, P301+P317, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P317, P321, P330, P337+P317, P362+P364, P370+P378, P403+P235, P501 |
| Flash point | −37 °C (−35 °F)[7] |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Tetramethylgermanium or tetramethylgermane is an organogermanium compound with the chemical formula Ge(CH3)4, often abbreviated as GeMe4, where Me stands for methyl. It is a colorless liquid. It is the simplest tetraorganogermane.
Synthesis
Tetramethylgermanium can be synthesized by reaction between germanium tetrachloride and a Grignard reagent methylmagnesium chloride in diethyl ether.[6]
- GeCl4 + 4 CH3MgCl → Ge(CH3)4 + 4 MgCl2
Tetramethylgermanium can also be synthesized by reaction between germanium tetrachloride and dimethylzinc or dimethylcadmium. The latter is more easily separated from the resulting product due to its higher boiling point. The reaction proceeds almost quantitatively at room temperature.[9]
- GeCl4 + 2 Zn(CH3)2 → Ge(CH3)4 + 2 ZnCl2
Tetramethylgermanium can be also synthesized by heating germanium with methyl chloride in the presence of copper.
- Ge + 4 CH3Cl + 2 Cu → Ge(CH3)4 + 2 CuCl2
Characteristics
Tetramethylgermanium is a colorless oily liquid with a sweetish odor, similar to chloroform. It dissolves without decomposition in ethanol and diethyl ether. It reacts slowly with nitric acid at 0 °C.[6]
Structure
The tetramethylgermanium molecule is tetrahedral at the germanium and carbon atoms. The germanium atom is connected to four methyl groups by a single bond in a tetrahedral fashion, thus, tetramethylgermanium is a heavy analog of neopentane, where the central carbon atom is replaced by germanium.
Uses
Tetramethylgermanium is useful as a NMR chemical shift reference compound[1] and in production of thin films composed of germanium element and germanium-containing polymers using plasma polymerization technique. The colorless polymer films formed from tetramethylgermanium were investigated by elemental analysis and infrared spectroscopy. Those polymers contain −CH3, −CH2−, Ge−CH3, Ge−O−C, and Ge−O−Ge groups and germanium element. Most Ge species present at the outermost layers of the films are oxidized by air, while the Ge species at the inner layers exist as germanium element.[2]
Tetramethylgermanium can be used as a methylating agent in transmetallation reactions. The following reaction should be done below 20 °C (68 °F):[10]
- Ge(CH3)4 + GaCl3 → (CH3)3GeCl + CH3GaCl2
Safety
Upon contact with eyes, tetramethylgermanium can cause serious eye damage and serious eye irritation. It is harmful in contact with skin, causing skin irritation. It is a neurotoxin.[1] It is flammable. Its vapors may form explosive mixtures with air. Vapors of tetramethylgermanium are heavier than air and may spread along floors. While it burns, it releases toxic and irritating smoke and gases containing germanium oxides, carbon monoxide and carbon dioxide.[7]
References
- ^ a b c d e f g Tetramethylgermanium from PubChem
- ^ a b Inagaki, N.; Mitsuuchi, M. (October 1983). "Glow discharge polymerization of tetramethylgermanium". Journal of Polymer Science: Polymer Chemistry Edition. 21 (10): 2887–2895. doi:10.1002/pol.1983.170211004.
- ^ "Tetramethylgermanium | Tetramethylgermane | C4H12Ge". Retrieved 2026-02-07.
- ^ "CAS 865-52-1 Tetramethylgermane - Alfa Chemistry". www.alfa-chemistry.com. Retrieved 2026-02-07.
- ^ a b "Tetramethylgermanium, 98% 25 g". Thermo Scientific Chemicals. Retrieved 2026-02-07.
- ^ a b c d Dennis, L. M. (2 August 1928). "Germanium. Zusammenfassung der Untersuchungen im Department of Chemistry, Cornell University, 1921–1927" [Germanium. Summary of investigations in the Department of Chemistry, Cornell University, 1921–1927]. Zeitschrift für anorganische und allgemeine Chemie (in German). 174 (1): 97–141. doi:10.1002/zaac.19281740114.
- ^ a b c d e "Tetramethylgermanium". Sigma-Aldrich. Retrieved February 13, 2026.
- ^ a b "Tetramethylgermane". ChemSpider. Retrieved 2026-02-07.
- ^ Long, L.H.; Pulford, C.I. (August 1968). "The preparation and physical properties of highly pure tetramethylgermane". Journal of Inorganic and Nuclear Chemistry. 30 (8): 2071–2075. doi:10.1016/0022-1902(68)80199-X.
- ^ Schmidbaur, H.; Findeiss, W. (October 1964). "A Simple Route to Organogallium Compounds". Angewandte Chemie International Edition in English. 3 (10): 696. doi:10.1002/anie.196406961.