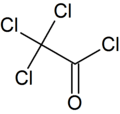
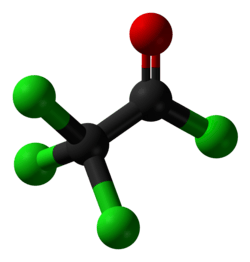
Trichloroacetyl chloride
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Trichloroacetyl chloride | |||
| Other names
2,2,2-Trichloroacetyl chloride
| |||
| Identifiers | |||
CAS Number
|
|||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.000.843 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
| UN number | 2442 | ||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
C2Cl4O | ||
| Molar mass | 181.832 g/mol | ||
| Density | 1.62 g/cm3 at 20 °C | ||
| Boiling point | 117.9 °C (244.2 °F; 391.0 K) | ||
| Solubility | miscible with diethyl ether[1] | ||
| Thermochemistry | |||
Std enthalpy of
formation (ΔfH⦵298) |
−280.0 kJ•mol−1[2] | ||
| Hazards | |||
| GHS labelling:[3] | |||
Pictograms
|
  
| ||
Signal word
|
Danger | ||
Hazard statements
|
H302, H314, H330 | ||
Precautionary statements
|
P260, P264, P270, P271, P280, P284, P301+P312, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P320, P330, P363, P403+P233, P405 | ||
| Safety data sheet (SDS) | Oxford MSDS | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
Trichloroacetyl chloride is the acyl chloride of trichloroacetic acid. It can be formed by reacting chlorine with acetyl chloride or acetaldehyde in the presence of activated charcoal, or by the isomerisation of tetrachloroethylene oxide via heating. It is used in the manufacture of pharmaceuticals and plant protection compounds.[4]
References
- ^ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, pp. 3–536, ISBN 0-8493-0594-2
- ^ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, pp. 5–29, ISBN 0-8493-0594-2
- ^ "Trichloroacetyl chloride". pubchem.ncbi.nlm.nih.gov. Retrieved 12 December 2021.
- ^ US Patent No. 5,659,078 to Ebmeyer et al., "Process for the preparation of trichloroacetyl chloride," issued August 19, 1997 (as reproduced by freepatentsonline.com and retrieved on October 23, 2007).