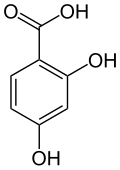
2,4-Dihydroxybenzoic acid
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2,4-Dihydroxybenzoic acid | |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.001.770 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties[2] | |
Chemical formula
|
C7H6O4 |
| Molar mass | 154.121 g·mol−1 |
| Melting point | 229 °C (444 °F; 502 K) |
Solubility in water
|
soluble |
| Solubility in ethanol | soluble |
| Solubility in diethyl ether | soluble |
| Solubility in benzene | soluble |
| Acidity (pKa) |
|
| Hazards | |
| GHS labelling:[3] | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H315, H319, H335 |
Precautionary statements
|
P261, P280, P302+P352, P305+P351+P338 |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
2,4-Dihydroxybenzoic acid (β-resorcylic acid) is a dihydroxybenzoic acid.
As a resorcylic acid, it is one of the three isomeric crystalline acids that are both carboxyl derivatives of resorcinol and dihydroxy derivatives of benzoic acid. Synthesis from resorcinol is via the Kolbe-Schmitt reaction.[4]
It is a degradation product of cyanidin glycosides from tart cherries in cell cultures.[5] It is also a metabolite found in human plasma after cranberry juice consumption.[6]
References
- ^ Haynes, William M., ed. (2016). "5". CRC Handbook of Chemistry and Physics (PDF) (97th ed.). CRC Press. p. 91. ISBN 9781498754293.
- ^ Haynes, William M., ed. (2016). "3". CRC Handbook of Chemistry and Physics (PDF) (97th ed.). CRC Press. p. 190. ISBN 9781498754293.
- ^ GHS: GESTIS 492493
- ^ Becker, Heinz G.O., ed. (1993). Organikum: organisch-chemisches Grundpraktikum (in German) (19th (rev. and expanded) ed.). Leipzig: Johann Ambrosius Barth. pp. 351–352. ISBN 978-3-335-00343-4.
- ^ Seeram, Navindra P.; Bourquin, Leslie D.; Nair, Muraleedharan G. (2001). "Degradation Products of Cyanidin Glycosides from Tart Cherries and Their Bioactivities". Journal of Agricultural and Food Chemistry. 49 (10): 4924–4929. doi:10.1021/jf0107508. PMID 11600045.
- ^ Zhang, Kai; Zuo, Yuegang (2004). "GC-MS Determination of Flavonoids and Phenolic and Benzoic Acids in Human Plasma after Consumption of Cranberry Juice". Journal of Agricultural and Food Chemistry. 52 (2): 222–227. doi:10.1021/jf035073r. PMID 14733499.