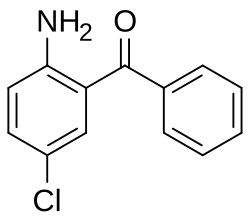
2-Amino-5-chlorobenzophenone
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(2-Amino-5-chlorophenyl)(phenyl)methanone | |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.010.864 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C13H10ClNO |
| Molar mass | 231.68 g·mol−1 |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H315, H319, H335 |
Precautionary statements
|
P261, P264, P264+P265, P271, P280, P302+P352, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
2-Amino-5-chlorobenzophenone is a substituted benzophenone that can be used in the synthesis of benzodiazepines.
Chemistry
2-Amino-5-chlorobenzophenone is a substituted derivative of benzophenone, where the hydrogen atom at the 2 position has been replaced by an amino group (-NH2) and the one at the 5 position replaced with a chlorine atom.[1]
Synthesis
Among other methods, 2-amino-5-chlorobenzophenone can be synthesized by reducing isoxazole through iron powder. This process also involves using toluene and muriatic acid.[2]
Synthesis of benzodiazepines
2-Amino-5-chlorobenzophenone and its derivatives can be used to produce benzodiazepines, a few examples are listed below;
Prazepam
Prazepam can be produced by the acylation of 2-amino-5-chlorobenzophenone with cyclo-propanecarbonyl chloride and triethylamine, 2-cyclopropylmethylamino-5-chlorobenzhydrol is then obtained by using lithium aluminium hydride as a reducing agent, this product is then oxidized by using manganese dioxide. The resulting compound goes another acylation reaction using phthalimidoacetyl chloride and finally treated with hydrazine hydrate to produce prazepam.[3]
Lorazepam
Lorazepam can be made using 2-amino-2′,5-dichlorobenzophenone (a derivative of 2-amino-5-chlorobenzophenone), which is first reacted with hydroxylamine, the obtained product is then reacted with chloroacetyl chloride to give 6-chloro-2-chlormethyl-4-(2′-chlorophenyl)quinazolin-3-oxide, a reaction with methylamine produces ring expansion and rearrangement, which forms 7-chloro-2-methylamino-5-(2′-chlorphenyl)-3H-1,4-benzodiazepin-4-oxide, acetylation with acetic anhydride gives a product which goes under hydrolysis by reacting it with hydrochloric acid, this gives 7-chloro-5-(2′-chlorophenyl)-1,2-dihydro-3H-1,4-benzodiazepin-2-on-4-oxide, a second reaction with acetic anhydride gives 7-chloro-1,3-dihydro-3-acetoxy-5-(2′-chlorphenyl)-2H-benzodiazepin-2-one, the last step involves hydrolysis of this product under sodium hydroxide to give lorazepam.[4]
Chlordiazepoxide
To make chlordiazepoxide, 2-amino-5-chlorobenzophenone is first reacted with hydroxylamine, the resulting product is then reacted with chloracetyl chloride in acetic acid, resulting in 6-chloro-2-chloromethyl-4-phenylquinazolin-3-oxide, reaction with methylamine gives chlordiazepoxide.[4]
References
- ^ a b c 2-Amino-5-chlorobenzophenone from PubChem
- ^ CN104230727A, 刘为焱; 潘明 & 黄雄, "Synthesis technology for producing 2-amino-5-chlorobenzophenone by reducing isoxazole through iron powder", issued 2014-12-24
- ^ "Prazepam". Some Pharmaceutical Drugs. IARC Monographs on the Evaluation of Carcinogenic Risks to Humans. Vol. 66. 1996. pp. 143–155. ISBN 978-92-832-1266-9. PMC 7681523. PMID 9097122.
- ^ a b Vardanyan, R.S.; Hruby, V.J. (2006). "Anxiolytics (Tranquilizers)". Synthesis of Essential Drugs. pp. 69–82. doi:10.1016/B978-044452166-8/50005-4. ISBN 978-0-444-52166-8.