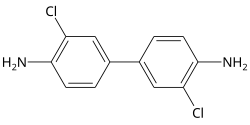
3,3'-Dichlorobenzidine
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
3,3′-Dichloro[1,1′-biphenyl]-4,4′-diamine | |
| Other names
4-(4-Amino-3-chlorophenyl)-2-chloroaniline
4,4′-Diamino-3,3′-dichlorobiphenyl o,o'-Dichlorobenzidine 3,3′-Dichlorobiphenyl-4,4′-diamine 3,3′-Dichloro-4,4′-biphenyldiamine 3,3′-Dichloro-4,4′-diaminobiphenyl | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.918 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C12H10Cl2N2 |
| Molar mass | 253.13 g/mol |
| Appearance | Gray or purple crystalline solid |
| Melting point | 132 to 133 °C (270 to 271 °F; 405 to 406 K) |
| Boiling point | 402 °C (756 °F; 675 K) |
Solubility in water
|
0.07% (15°C)[2] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Potential carcinogen[2] |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
carcinogen[2] |
REL (Recommended)
|
Ca[2] |
IDLH (Immediate danger)
|
Ca [N.D.][2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
3,3'-Dichlorobenzidine is a chlorinated aromatic amine derived from benzidine. It is widely used in the production of diarylide yellow pigments used in the production of printing inks.[3] Its use in the production of dyes has been largely discontinued because of concerns about carcinogenicity.
Properties and the market
3,3'-Dichlorobenzidine is a crystalline solid soluble in alcohol, benzene, diethyl ether, and glacial acetic acid, but difficultly soluble in dilute hydrochloric acid and water.[4] It exists in gray-to-purple needle-like crystals.[4] Commercial powder samples may also be light-brown or violet.[5]
This compound is often confused with its dihydrochloride salt, even in scientific literature.[4] 3,3'-Dichlorobenzidine dihydrochloride is sparingly soluble in water and readily soluble in alcohol. It is marketed in the form of a colorless to pale gray crystalline powder. Both the commercial 3,3'-dichlorobenzidine and its hydrochloride usually contain 5 to 30% of water.[5]
The so-called urethane quality 3,3'-dichlorobenzidine is anhydrous and free of hydrochloric acid.[5] However, some sources dispute the very existence of the free base on the market and believe the dihydrochloride salt to be the only form available commercially.[4]
Preparation and reactions
3,3'-Dichlorobenzidine is prepared in two steps from 2-nitrochlorobenzene. The first step involves reduction with zinc in base to afford 2,2'-dichlorodiphenylhydrazine. This intermediate undergoes the benzidine rearrangement to afford 3,3'-dichlorobenzidine.[5]
Aqueous solutions of 3,3'-dichlorobenzidine degrade in light to monochloro derivative. It undergoes chlorination (for example in water treatment plants) to give the tetrachloro derivative.
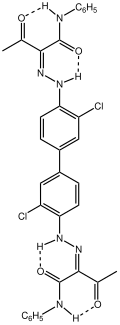
The most widely practiced reaction of 3,3'-dichlorobenzidine is its double diazotization. This bis(diazo) intermediate is then coupled to derivatives of acetoacetylaminobenzene (CH3C(O)CH2C(O)NHAr). In this way, the diarylide commercial yellow pigments are produced: Pigment Yellow 12, Pigment Yellow 13, Pigment Yellow 14, Pigment Yellow 17, Pigment Orange 13, Pigment Yellow 81, and Pigment Yellow 83.[3]
Safety
3,3'-Dichlorobenzidine is considered a carcinogen.[1] This compound has been shown to increase the incidence of tumors in animals.[7] Because it is structurally similar to benzidine, a known carcinogen, it is believed that it may share a similar mechanism in causing bladder cancer in humans.[7]
References
- ^ a b Dichlorobenzidine - Compound Summary, PubChem.
- ^ a b c d e NIOSH Pocket Guide to Chemical Hazards. "#0191". National Institute for Occupational Safety and Health (NIOSH).
- ^ a b K. Hunger; W. Herbst (2012). "Pigments, Organic". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a20_371. ISBN 9783527303854.
- ^ a b c d "3,3′-Dichlorobenzidine and Its Dihydrochloride" (PDF). National Toxicology Program, Department of Health and Human Services. Retrieved 6 December 2025.
- ^ a b c d Schwenecke, H.; Mayer, D. (2005). "Benzidine and Benzidine Derivatives". Ullmann’s Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_539. ISBN 9783527303854.
- ^ Barrow, Michael J.; Christie, Robert M.; Lough, Alan J.; Monteith, Jean E.; Standring, Paul N. (2000). "The Crystal Structure of C.I. Pigment Yellow 12". Dyes and Pigments. 45 (2): 153–160. doi:10.1016/S0143-7208(00)00017-6.
- ^ a b "3, 3'-Dichlorobenzidine". U.S. Environmental Protection Agency, Integrated Risk Information System. 7 March 2011. Accessed 3 May 2011.