Aluminium antimonide
 | |
| Identifiers | |
|---|---|
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.042.410 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
AlSb |
| Molar mass | 148.742 g/mol |
| Appearance | black crystals |
| Density | 4.26 g/cm3 |
| Melting point | 1,060 °C (1,940 °F; 1,330 K) |
| Boiling point | 2,467 °C (4,473 °F; 2,740 K) |
Solubility in water
|
insoluble |
| Band gap | 1.58 eV |
Refractive index (nD)
|
3.3 |
| Structure | |
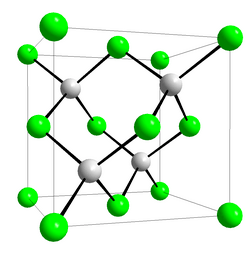
Crystal structure
|
Zinc blende |
Space group
|
T2d-F-43m |
Coordination geometry
|
Tetrahedral |
| Thermochemistry | |
Std molar
entropy (S⦵298) |
65 J/mol K |
Std enthalpy of
formation (ΔfH⦵298) |
−50.4 kJ/mol |
| Hazards | |
| NFPA 704 (fire diamond) | 
0
3
1 |
Autoignition
temperature |
317 °C (603 °F; 590 K) |
| Safety data sheet (SDS) | MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Aluminium antimonide (AlSb) is a semiconductor of the group III-V family containing aluminium and antimony. The lattice constant is 0.61 nm. The indirect bandgap is approximately 1.6 eV at 300 K, whereas the direct band gap is 2.22 eV.
Its electron mobility is 200 cm2·V−1·s−1 and hole mobility 400 cm2·V−1·s−1 at 300 K. Its refractive index is 3.3 at a wavelength of 2 μm, and its dielectric constant is 10.9 at microwave frequencies.[1]
AlSb can be reacted with other III-V materials to produce ternary materials including AlInSb, AlGaSb and AlAsSb.
Aluminium antimonide is rather flammable because of the reducing tendency of the antimonide (Sb3−) ion. It burns to produce aluminium oxide and antimony trioxide.
See also
References
- ^ K Seeger and E Schonherr "Microwave dielectric constant of aluminium antimonide" Semicond. Sci. Technol. 6 (1991) 301 doi:10.1088/0268-1242/6/4/013