Beryllium bromide
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Beryllium bromide
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.196 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
BeBr2 |
| Molar mass | 168.820 g/mol |
| Appearance | colorless white crystals |
| Density | 3.465 g/cm3 (20 °C) |
| Melting point | 508 °C (946 °F; 781 K)sublimes at 473 °C (883 °F; 746 K) |
| Boiling point | 520 °C (968 °F; 793 K)[1] |
Solubility in water
|
Highly[1] |
| Solubility | soluble in ethanol, diethyl ether, pyridine insoluble in benzene |
| Structure | |
Crystal structure
|
Orthorhombic |
| Thermochemistry | |
Heat capacity (C)
|
0.4111 J/g K |
Std molar
entropy (S⦵298) |
9.5395 J/K |
Std enthalpy of
formation (ΔfH⦵298) |
−2.094 kJ/g |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
see Berylliosis |
| GHS labelling: | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H301, H315, H317, H319, H330, H335, H350i, H372, H411 |
Precautionary statements
|
P260, P301+P310, P304+P340, P305+P351+P338, P320, P330, P405, P501 |
| NFPA 704 (fire diamond) | 
4
0
0 |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 0.002 mg/m3 C 0.005 mg/m3 (30 minutes), with a maximum peak of 0.025 mg/m3 (as Be)[2] |
REL (Recommended)
|
Ca C 0.0005 mg/m3 (as Be)[2] |
IDLH (Immediate danger)
|
Ca [4 mg/m3 (as Be)][2] |
| Related compounds | |
Other anions
|
Beryllium fluoride Beryllium chloride Beryllium iodide |
Other cations
|
Magnesium bromide Calcium bromide Strontium bromide Barium bromide Radium bromide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Beryllium bromide is the chemical compound with the formula BeBr2. It is very hygroscopic and dissolves well in water. The Be2+ cation, which is relevant to BeBr2, is characterized by the highest known charge density (Z/r = 6.45), making it one of the hardest cations and a very strong Lewis acid.[3]
Preparation and reactions
It can be prepared by reacting beryllium metal with elemental bromine at temperatures of 500 °C to 700 °C:[1]
- Be + Br2 → BeBr2
When the oxidation is conducted on an ether suspension, one obtains colorless dietherate:[4]
- Be + Br2 + 2 O(C2H5)2 → BeBr2(O(C2H5)2)2
The same dietherate is obtained by suspending beryllium dibromide in diethyl ether:[5]
- BeBr2 + 2 O(C2H5)2 → BeBr2(O(C2H5)2)2
This ether ligand can be displaced by other Lewis bases
Beryllium bromide hydrolyzes slowly in water:
BeBr2 + 2 H2O → 2 HBr + Be(OH)2
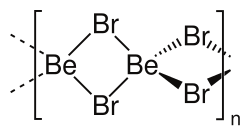
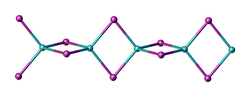
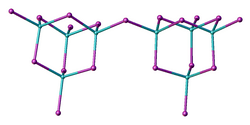
Structure
Two forms (polymorphs) of BeBr2 are known. Both structures consist of tetrahedral Be2+ centers interconnected by doubly bridging bromide ligands. One form consist of edge-sharing polytetrahedra. The other form resembles zinc iodide with interconnected adamantane-like cages.[6][7]
Safety
Beryllium compounds are toxic if inhaled or ingested.
References
- ^ a b c Perry, Dale L.; Phillips, Sidney L. (1995), Handbook of Inorganic Compounds, CRC Press, pp. 61–62, ISBN 0-8493-8671-3, retrieved 2007-12-10
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0054". National Institute for Occupational Safety and Health (NIOSH).
- ^ Buchner, M. R. (2017-01-01), "Beryllium Chemistry", Reference Module in Chemistry, Molecular Sciences and Chemical Engineering, Elsevier, ISBN 978-0-12-409547-2, retrieved 2022-10-27
{{citation}}: CS1 maint: work parameter with ISBN (link) - ^ Paparo, Albert; Jones, Cameron (2019-02-01). "Beryllium Halide Complexes Incorporating Neutral or Anionic Ligands: Potential Precursors for Beryllium Chemistry". Chemistry: An Asian Journal. 14 (3): 486–490. doi:10.1002/asia.201801800. ISSN 1861-4728. PMID 30604490. S2CID 58632466.
- ^ Paparo, Albert; Jones, Cameron (2019-02-01). "Beryllium Halide Complexes Incorporating Neutral or Anionic Ligands: Potential Precursors for Beryllium Chemistry". Chemistry: An Asian Journal. 14 (3): 486–490. doi:10.1002/asia.201801800. ISSN 1861-4728. PMID 30604490. S2CID 58632466.
- ^ Buchner, Magnus R.; Dankert, Fabian; Spang, Nils; Pielnhofer, Florian; von Hänisch, Carsten (2020). "A Second Modification of Beryllium Bromide: β-BeBr2". Inorganic Chemistry. 59 (23): 16783–16788. doi:10.1021/acs.inorgchem.0c02832. PMID 33185106. S2CID 226850424.
- ^ Troyanov, S. I. (2000). "Crystal Modifications of Beryllium Dihalides BeCl2, BeBr2, and BeI2". Zhurnal Neorganicheskoi Khimii. 45: 1619-1624.