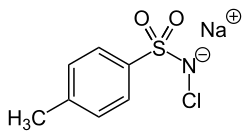
Chloramine-T
 | |
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium chloro(4-methylbenzene-1-sulfonyl)azanide | |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.414 |
| EC Number |
|
| KEGG | |
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C7H7ClNO2S·Na C7H7ClNO2S·Na·(3H2O) (hydrate) |
| Molar mass | 227.64 g/mol 281.69 g/mol (trihydrate) |
| Appearance | White powder |
| Density | 1.4 g/cm3 |
| Melting point | Releases chlorine at 130 °C (266 °F; 403 K) Solid melts at 167–169 °C |
Solubility in water
|
>100 g/L (hydrate)[1] |
| Pharmacology | |
ATC code
|
D08AX04 (WHO) QP53AB04 (WHO) |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Corrosive |
| GHS labelling: | |
Pictograms
|
|
Signal word
|
Danger |
Hazard statements
|
H302, H314, H334 |
Precautionary statements
|
P260, P264, P270, P280, P285, P301+P312, P301+P330+P331, P303+P361+P353, P304+P340, P304+P341, P305+P351+P338, P310, P321, P330, P342+P311, P363, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Chloramine-T is the organic compound with the formula CH3C6H4SO2NClNa. Both the anhydrous salt and its trihydrate are known. Both are white powders. Chloramine-T is used as a reagent in organic synthesis.[2] It is commonly used as cyclizing agent in the synthesis of aziridine, oxadiazole, isoxazole and pyrazoles.[3] It's inexpensive, has low toxicity and acts as a oxidizing agent. In addition, it also acts as a source of nitrogen anions and electrophilic cations. It may undergo degradation on long term exposure to atmosphere such that care must be taken during its storage.
Reactions
Chloramine-T contains active (electrophilic) chlorine. Its reactivity is similar to that of sodium hypochlorite. Aqueous solutions of chloramine-T are slightly basic (pH typically 8.5). The pKa of the closely related N-chlorophenylsulfonamide C6H5SO2NClH is 9.5.[2]
It is prepared by oxidation of toluenesulfonamide with sodium hypochlorite, with the latter being produced in situ from sodium hydroxide and chlorine (Cl2):[2]

Uses
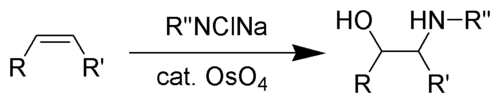
Reagent in amidohydroxylation
The Sharpless oxyamination converts an alkene to a vicinal aminoalcohol. A common source of the amido component of this reaction is chloramine-T.[4] Vicinal aminoalcohols are important products in organic synthesis and recurring pharmacophores in drug discovery.
Oxidant
Chloramine-T is a strong oxidant. It oxidizes hydrogen sulfide to sulfur, and mustard gas (bis(2-chloroethyl) sulfide) to yield a harmless crystalline sulfimide.[5]
It converts iodide to iodine monochloride (ICl). ICl rapidly undergoes electrophilic substitution predominantly with activated aromatic rings, such as those of the amino acid tyrosine. This makes it a useful reagent in combination with an iodide ion source for iodination of peptides and proteins. Chloramine-T together with iodogen (1,3,4,6-Tetrachloro-3a,6a-diphenyltetrahydroimidazo[4,5-d]imidazole-2,5(1H,3H)-dione) or lactoperoxidase is commonly used for labeling peptides and proteins with radioiodine isotopes.[6]
Disinfectant
Chloramine-T has a long history as a hospital disinfectant. It is effective against e.g. hepatitis and HI viruses.[7] Unlike the more common sodium hypochlorite, chloramine-T is mildly basic, almost odorless and is not a bleaching agent.[8]
Safety
Chloramine-T is harmful if swallowed. It is corrosive on skin, eyes or mucous membranes. It releases toxic chlorine gas upon reaction with acids. It is water-soluble and thus can be released to the environment dissolved in water. It is a known sensitizer.[9] Chloramine-T has been observed to cause occupational asthma and flu-like symptoms.[7][10]
Certifications
- EN 1276 Bactericidal
- EN 13713 Bactericidal
- EN 14675 Virucidal
- EN 14476 Virucidal Norovirus
- EN 1650 Fungicidal
- EN 13704 Sporicidal Clostridioides difficile
References
- ^ "Chloramine-T hydrate". Sigma-Aldrich.
- ^ a b c Campbell, Malcolm M.; Johnson, Graham. (1978). "Chloramine T and Related N-halogeno-N-metallo reagents". Chemical Reviews. 78: 65–79. doi:10.1021/cr60311a005.
- ^ Nayak, Yogeesha N.; Gaonkar, Santosh L.; Saleh, Ebraheem Abdu Musad; Dawsari, Abdullah Mohammed A. L.; Harshitha; Husain, Kakul; Hassan, Ismail (2022-03-01). "Chloramine-T (N-chloro-p-toluenesulfonamide sodium salt), a versatile reagent in organic synthesis and analytical chemistry: An up to date review". Journal of Saudi Chemical Society. 26 (2) 101416. doi:10.1016/j.jscs.2021.101416. ISSN 1319-6103.
- ^ Bodkin, J. A.; McLeod, M. D. (2002). "The Sharpless asymmetric aminohydroxylation". J. Chem. Soc., Perkin Trans. 1. 2002 (24): 2733–2746. doi:10.1039/b111276g.
- ^ Ura, Yasukazu; Sakata, Gozyo (2007). "Chloroamines". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a06_553. ISBN 978-3-527-30673-2.
- ^ Rösch, F. Radiochemistry and Radiopharmaceutical Chemistry in Life Sciences. Vol. 4. Dordrecht, Boston, London: Kluwer Academic Publishers.
- ^ a b "Kloramiini-T-jauheesta ammattiastmaa ja nuhaa välinehuoltotyössä" [Chloramine-T-powder relieves occupational asthma and rhinitis in equipment maintenance work] (in Finnish). February 23, 1995. Retrieved December 27, 2025.
{{cite web}}: CS1 maint: url-status (link) - ^ "Sactiv Kloramiini 1l". www.sillasiisti.fi (in Finnish). Retrieved 2024-10-25.
- ^ https://www.carlroth.com/medias/SDB-0271-GB-EN.pdf?context=bWFzdGVyfHNlY3VyaXR5RGF0YXNoZWV0c3wzMDc2NDF8YXBwbGljYXRpb24vcGRmfGgwYi9oZjEvOTE0MzI0MzgwMDYwNi9TREJfMDI3MV9HQl9FTi5wZGZ8NzE3ODFiZjc3YjFlZTk1MWExMjUxOWU2YzJhOWEyZGY5ZDIxMzJjYTQ2NzU2MjI0MDBjMzVjZDMwMWJlODkyMQ
- ^ https://www.ttl.fi/file-download/download/public/6517
External links
- M. Shetty, T. B. Gowda (2004). "A Study of Substituent Effect on the Oxidative Strengths of N-Chloroarenesulphonamides: Kinetics of Oxidation of Leucine and Isoleucine in Aqueous Acid Medium". Zeitschrift für Naturforschung. 59: 63–72. doi:10.1515/znb-2004-0110. S2CID 46154131.
- Chemicalland21.com: Chloramine T (Tosylchloramide sodium)
- InChem.org: Chloramine T
- "Disifin USA". Archived from the original on 2009-12-25. Retrieved 2010-02-09.