Gallium(III) fluoride
 | |
 | |
| Names | |
|---|---|
| Other names
gallium trifluoride
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.029.094 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
GaF3 |
| Molar mass | 126.718 g/mol |
| Appearance | white powder |
| Density | 4.47 g/cm3 |
| Melting point | 800 °C (1,470 °F; 1,070 K) |
| Boiling point | 1,000 °C (1,830 °F; 1,270 K) |
Solubility in water
|
0.0002 g/100 mL |
| Structure | |
Crystal structure
|
Rhombohedral, hR24 |
Space group
|
R-3c, No. 167 |
| Hazards | |
| GHS labelling: | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H302, H312, H332 |
Precautionary statements
|
P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P312, P304+P340, P312, P322, P330, P363, P501 |
| NFPA 704 (fire diamond) | 
3
0
0 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
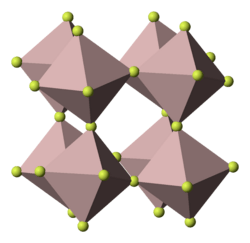
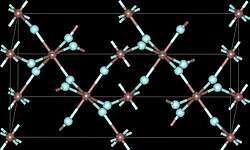
Gallium(III) fluoride (GaF3) is a chemical compound. It is a white solid that melts under pressure above 1000 °C but sublimes around 950 °C. It has the FeF3 structure where the gallium atoms are 6-coordinate.[1] GaF3 can be prepared by reacting F2 or HF with Ga2O3 or by thermal decomposition of (NH4)3GaF6.[2] GaF3 is virtually insoluble in water.[2] Solutions of GaF3 in HF can be evaporated to form the trihydrate, GaF3·3H2O, which on heating gives a hydrated form of GaF2(OH).[2] Gallium(III) fluoride reacts with mineral acids to form hydrofluoric acid.
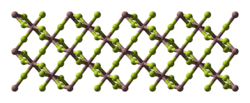 |
 |
 |
|
| view along the a axis | view along the c axis | Ga coordination | F coordination |
References
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.
- ^ a b c Anthony John Downs, (1993), Chemistry of Aluminium, Gallium, Indium, and Thallium, Springer, ISBN 978-0-7514-0103-5
Further reading
- Barrière, A.S.; Couturier, G.; Gevers, G.; Guégan, H.; Seguelond, T.; Thabti, A.; Bertault, D. (1989). "Preparation and characterization of gallium(III) fluoride thin films". Thin Solid Films. 173 (2): 243. Bibcode:1989TSF...173..243B. doi:10.1016/0040-6090(89)90140-5.