Caproic acid
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Hexanoic acid
| |
| Preferred IUPAC name
Hexanoic acid | |
| Other names
Hexoic acid
Hexylic acid Butylacetic acid Pentylformic acid 1-Pentanecarboxylic acid C6:0 (Lipid numbers) | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
773837 |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.005.046 |
| EC Number |
|
Gmelin Reference
|
185066 |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C6H12O2 |
| Molar mass | 116.160 g·mol−1 |
| Appearance | Oily liquid[1] |
| Odor | goat-like |
| Density | 0.929 g/cm3[2] |
| Melting point | −3.4 °C (25.9 °F; 269.8 K)[1] |
| Boiling point | 205.8 °C (402.4 °F; 478.9 K)[1] |
Solubility in water
|
1.082 g/100 mL[1] |
| Solubility | soluble in ethanol, ether |
| Acidity (pKa) | 4.88 |
Magnetic susceptibility (χ)
|
−78.55·10−6 cm3/mol |
Refractive index (nD)
|
1.4170 |
| Viscosity | 3.1 mP |
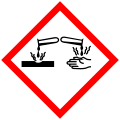
| Hazards | |
| GHS labelling: | |
Pictograms
|
|
Signal word
|
Danger |
Hazard statements
|
H314 |
Precautionary statements
|
P260, P264, P280, P301+P330+P331, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P310, P312, P321, P322, P361, P363, P405, P501 |
| NFPA 704 (fire diamond) | 
3
1
0 |
| Flash point | 103 °C (217 °F; 376 K)[2] |
Autoignition
temperature |
380 °C (716 °F; 653 K) |
| Explosive limits | 1.3-9.3% |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
3000 mg/kg (rat, oral) |
| Related compounds | |
Related compounds
|
Pentanoic acid, Heptanoic acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Caproic acid, also known as hexanoic acid, is the carboxylic acid derived from hexane with the chemical formula CH3(CH2)4COOH. It is a colorless oily liquid with a fatty, cheesy, waxy odor resembling that of goats[1] or other barnyard animals. It is a fatty acid found naturally in various animal fats and oils, and is one of the chemicals that gives the decomposing fleshy seed coat of the ginkgo its characteristic unpleasant odor.[3] It is also one of the components of vanilla and cheese. Salts and esters of caproic acid are known as caproates or hexanoates.
Two other acids are named after goats: caprylic acid (C8) and capric acid (C10). Along with caproic acid, they account for 15% of the fat in goat's milk.
Caproic, caprylic, and capric acids (capric is a crystal- or wax-like substance, whereas the other two are mobile liquids) are used for the formation of esters, and also commonly used "neat" in: butter, milk, cream, strawberry, bread, beer, nut, and other flavors.
Properties
Caproic acid is a 6-Carbon saturated fatty acid. It occurs in a white crystalline solid form or a colorless to pale yellow liquid state, accompanied by a strong and unpleasant odor. It has limited solubility in water, although it is soluble in dimethyl ether, benzene and other organic solvents.[4][5]
It may be prepared by fractionation of the volatile fatty acids of coconut oil.[6] Caproic acid belongs to the family of medium chain fatty acids (MCFAs) which can be used to synthesize Medium-chain triglycerides.
Uses
The primary use of caproic acid is in the manufacture of its esters for use as artificial flavors, and in the manufacture of hexyl derivatives, such as hexylphenols.[1] Several progestin medications are caproate esters, such as hydroxyprogesterone caproate and gestonorone caproate. Its derivatives are also used in soaps, shampoos, conditioners and gels.
Caproic acid is also used as a component in varnish driers, as well as a lubricant. Its derivatives can also be incorporated to modify the elasticity, strength, and resilience of rubber products.[7]
DHEA-caproate ester (#121) has a IC50 of 0.049nM for the 5-alpha-reductase enzyme.[8][9]
See also
- List of carboxylic acids
- List of saturated fatty acids
- Caproate fermentation
References
- ^ a b c d e f The Merck Index: An Encyclopedia of Chemicals, Drugs, and Biologicals (11th ed.). Merck. 1989. ISBN 091191028X.
- ^ a b Record in the GESTIS Substance Database of the Institute for Occupational Safety and Health
- ^ "Ginkgo.html". Archived from the original on 2008-12-26. Retrieved 2007-03-08.
- ^ "What is Caproic Acid? Cosmetic usage, alternatives, and regulatory insights". Slate. Retrieved 2026-01-07.
- ^ PubChem. "Hexanoic Acid". pubchem.ncbi.nlm.nih.gov. Retrieved 2026-01-07.
- ^ "Hexanoic acid | 142-62-1". amp.chemicalbook.com. Retrieved 2026-01-07.
- ^ "Industrial Applications of Hexanoic Acid (CAS 142-62-1)". www.nbinno.com. Retrieved 2026-01-07.
- ^ Salvador, Jorge A.R.; Pinto, Rui M.A.; Silvestre, Samuel M. . (2013). Steroidal 5α-reductase and 17α-hydroxylase/17,20-lyase (CYP17) inhibitors useful in the treatment of prostatic diseases. The Journal of Steroid Biochemistry and Molecular Biology, 137(), 199–222. doi:10.1016/j.jsbmb.2013.04.006
- ^ Yazmín Arellano; Eugene Bratoeff; Mariana Garrido; Juan Soriano; Yvonne Heuze; Marisa Cabeza. (2011). New ester derivatives of dehydroepiandrosterone as 5α-reductase inhibitors. , 76(12), 0–1246. doi:10.1016/j.steroids.2011.05.015