Nitronium tetrafluoroborate
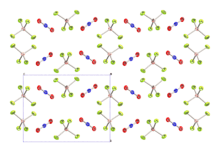
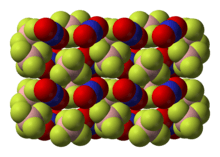
Nitronium tetrafluoroborate is an inorganic compound with formula NO2BF4. It is a salt of nitronium cation and tetrafluoroborate anion. It is a colorless crystalline solid, which reacts with water to form the corrosive acids HF and HNO3. As such, it must be handled under water-free conditions. It is sparsely soluble in many organic solvents.
 | |
 | |
| Names | |
|---|---|
| Other names
nitronium fluoroborate, NO 2BF 4 | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.034.107 |
| EC Number |
|
PubChem CID |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
BNO2F4 |
| Molar mass | 132.81 |
| Hazards | |
| Safety data sheet | |
| GHS pictograms | 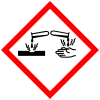   |
| GHS Signal word | Danger |
GHS hazard statements |
H314, H317, H334 |
GHS precautionary statements |
P260, P261, P264, P272, P280, P285, P301+330+331, P302+352, P303+361+353, P304+340, P304+341, P305+351+338, P310, P321, P333+313, P342+311, P363, P405, P501 |
| Related compounds | |
Other cations |
Nitrosonium tetrafluoroborate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Preparation
Nitronium tetrafluoroborate can be prepared by adding a mixture of anhydrous hydrogen fluoride and boron trifluoride to a nitromethane solution of nitric acid or dinitrogen pentoxide.[1]
Applications
Nitronium tetrafluoroborate is used as a nitration agent.
References
- Kenneth Schofield (1980). Aromatic nitration. CUP Archive. p. 88. ISBN 0-521-23362-3.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.