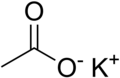
Potassium acetate
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Potassium acetate | |||
| Other names
Potassium ethanoate
| |||
| Identifiers | |||
CAS Number
|
|||
3D model (JSmol)
|
|||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.004.385 | ||
| E number | E261 (preservatives) | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
CH3COOK | ||
| Molar mass | 98.142 g·mol−1 | ||
| Appearance | White deliquescent crystalline powder | ||
| Density | 1.57 g/cm3 (25 °C (77 °F; 298 K))[1] | ||
| Melting point | 303–307 °C (577–585 °F; 576–580 K)[1] | ||
| Boiling point | Decomposes | ||
Solubility in water
|
| ||
| Solubility in methanol |
| ||
| Solubility in ethanol | 16.3 g/100g[2] | ||
| Solubility in acetic acid | 20.95 g/100g (28.03 °C (82.45 °F; 301.18 K))[3][4] | ||
| Solubility in dimethylformamide | 0.09 g/100g[3] | ||
| Solubility in sulfur dioxide | 0.006 g/100g (0 °C (32 °F; 273 K))[3] | ||
| Solubility in liquid ammonia | 1.026 g/100g (−33.9 °C (−29.0 °F; 239.2 K))[3] | ||
| Vapor pressure | < 0.0000001 hPa[1] | ||
| Acidity (pKa) | 4.76 | ||
| Structure | |||
Crystal structure
|
Monoclinic | ||
| Thermochemistry[5] | |||
Heat capacity (C)
|
109.38 J/(mol × K) | ||
Std molar
entropy (S⦵298) |
150.82 J/(mol × K) | ||
Std enthalpy of
formation (ΔfH⦵298) |
−722.6 kJ⋅mol−1[3] | ||
Enthalpy of fusion (ΔfH⦵fus)
|
22 kJ⋅mol−1[3] | ||
| Pharmacology | |||
ATC code
|
B05XA17 (WHO) | ||
| Hazards[6] | |||
| GHS labelling: | |||
Signal word
|
Warning | ||
| NFPA 704 (fire diamond) | 
1
1
1 | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
3250 mg/kg (oral, rat) | ||
| Related compounds | |||
Other anions
|
| ||
Other cations
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
Potassium acetate (also called potassium ethanoate), (CH3COOK) is the potassium salt of acetic acid. It is a hygroscopic solid at room temperature.
Preparation
It can be prepared by treating a potassium-containing base such as potassium hydroxide with acetic acid:
- CH3COOH + KOH → CH3COOK + H2O
This sort of reaction is known as an acid-alkali reaction.
At saturation, the sesquihydrate in water solution (2CH3COOK·3H2O) begins to form the semihydrate (2CH3COOK·H2O) at 41.3 °C (106.3 °F).[2]
Applications
Deicing
Potassium acetate (as a substitute for calcium chloride or magnesium chloride) can be used as a deicer to remove ice or prevent its formation. It offers the advantages over chlorides of being less aggressive on soils and much less corrosive: for this reason, it is one of the preferred substances for removal of ice from airplanes and airport runways.[7][8]
Fire extinguishing
Potassium acetate is the extinguishing agent used as a component in some Class K fire extinguishers[9] because of its ability to cool and form a crust over burning oils.
Food additive
Potassium acetate is used in processed foods as a preservative and acidity regulator. In the European Union, it is labeled by the E number E261; it is also approved for usage in the USA, Australia, and New Zealand.[10][11][12]
Medicine and biochemistry
In molecular biology, potassium acetate is used to precipitate Sodium dodecyl sulfate (SDS) and SDS-bound proteins to allow their removal from DNA.
Potassium acetate is used in mixtures applied for tissue preservation, fixation, and mummification. Most museums today use a formaldehyde-based method recommended by Kaiserling in 1897 which contains potassium acetate.[13] This process was used to soak Lenin's corpse.[14]
Industry
Potassium acetate is used as a catalyst in the production of polyurethanes.[15]
Use in executions
Potassium acetate was incorrectly used in place of potassium chloride when putting a prisoner to death in Oklahoma in January 2015. Charles Frederick Warner was executed on January 15, 2015 with potassium acetate; this was not public knowledge until the scheduled execution of Richard Glossip was called off.[16][17]
In August 2017, the U.S. state of Florida used potassium acetate (intentionally) in the execution of Mark James Asay.[18]
Historical
Potassium acetate is used as a diuretic and urinary alkalizer. Before modern chemistry, it was variously called terra foliata tartari, sal Sennerti, tartarus regeneratus, arcanum tartari and sal diureticus.[19] In 1760 it was used in the preparation of Cadet's fuming liquid ((CH3)2As)2 + ((CH3)2As)2O, the first organometallic compound ever produced.
References
- ^ a b c Sigma-Aldrich Co., Potassium acetate.
- ^ a b c Seidell, Atherton; Linke, William F. (1919). "Potassium - Potassium acetate". Solubilities of Inorganic and Organic Compounds (2nd ed.). New York: Van Nostrand. p. 500. Retrieved 31 October 2025.
- ^ a b c d e f g "Potassium acetate". chemister.ru. Chemister. Retrieved 31 October 2025.
- ^ Davidson, Arthur W.; McAllister, Walter H. (1 February 1930). "Solutions of salts in pure acetic acid. II. Solubilities of acetates". Journal of the American Chemical Society. 52 (2): 507–519. Bibcode:1930JAChS..52..507D. doi:10.1021/ja01365a010.
- ^ Acetic acid, potassium salt in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD) (retrieved 2014-05-18)
- ^ "SDS - Potassium acetate". fishersci.com. Thermo Fisher Scientific. Retrieved 31 October 2025.
- ^ Gruber, Michael R.; Hofko, Bernhard; Hoffmann, Markus; Stinglmayr, David; Seifried, Teresa M.; Grothe, Hinrich (April 2023). "Deicing performance of common deicing agents for winter maintenance with and without corrosion-inhibiting substances". Cold Regions Science and Technology. 208 103795. doi:10.1016/j.coldregions.2023.103795.
- ^ Chappelo, C.C.; McElroy, A.D.; Blackburn, R.R.; Darwin, D.; de Noyelles, F.G.; Locke, C.E. (1992). Handbook of test methods for evaluating chemical deicers (PDF). Washington, DC: Strategic Highway Research Program, National Research Council. p. 73. ISBN 9789994805839. SHRP-H-332.
- ^ "SDS - Class K liquid agent for extinguishers" (PDF). Amerex. May 2016. p. 2. Retrieved 31 October 2025.
- ^ "Current EU approved additives and their E Numbers". UK Food Standards Agency. Retrieved 2011-10-27.
- ^ "Listing of Food Additives Status Part II". Food and Drug Administration. US Food and Drug Administration. Archived from the original on January 8, 2010. Retrieved 2011-10-27.
- ^ "Standard 1.2.4 - Labelling of ingredients". Australia-New Zealand Food Standards Code. 8 September 2011. Retrieved 2025-06-14.
- ^ Dale Ulmer (1994). "Fixation. The Key to Good Tissue Preservation" (PDF). Journal of the International Society for Plastination. 8 (1): 7–10. doi:10.56507/OEVY7072. Archived from the original (PDF) on 2011-05-16. Retrieved 2017-09-08.
- ^ Nagorski, Andrew (18 September 2007). The Greatest Battle: Stalin, Hitler, and the Desperate Struggle for Moscow That Changed the Course of World War II (1st ed.). Simon and Schuster. p. 53. ISBN 978-0743281102.
- ^ Cheung, H.; Tanke, R. S.; Torrence, G. P. "Acetic Acid". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a01_045. ISBN 978-3-527-30673-2.
- ^ Gajanan, Mahita (8 October 2015). "Oklahoma used wrong drug in Charles Warner's execution, autopsy report says". The Guardian.
- ^ Peralta, Eyder (8 October 2015). "Oklahoma Used The Wrong Drug To Execute Charles Warner". NPR.
- ^ Dearon, Jason (3 June 2018) [24 August 2017]. "Florida executes convicted killer Mark Asay using new drug". Sun Sentinel.
- ^ Crosland, Maurice P. (2004-01-01). Historical Studies in the Language of Chemistry. Courier Corporation. ISBN 978-0-486-43802-3.