Rhenium(IV) oxide
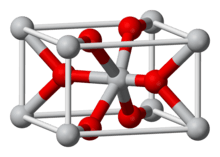
Rhenium(IV) oxide or rhenium dioxide is the inorganic compound with the formula ReO2. This gray to black crystalline solid is a laboratory reagent that can be used as a catalyst. It adopts the rutile structure.
 | |
| Names | |
|---|---|
| IUPAC name
Rhenium(IV) oxide | |
| Other names
Rhenium dioxide | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
| ECHA InfoCard | 100.031.659 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
ReO2 |
| Molar mass | 218.206 g/mol |
| Appearance | gray orthorhombic crystals |
| Density | 11.4 g/cm3[1] |
| Melting point | decomposes at 1000 °C[2] |
Solubility in water |
insoluble |
| Solubility in alkali | insoluble |
Magnetic susceptibility (χ) |
+44.0·10−6 cm3/mol |
| Structure | |
Crystal structure |
Orthorohmbic, oP12 |
Space group |
Pbcn, No. 60 |
| Hazards | |
| Safety data sheet | Aldrich MSDS |
| NFPA 704 (fire diamond) | 
0
1
0 |
| Related compounds | |
Other anions |
Rhenium(VII) oxide Rhenium(III) oxide Rhenium(III) chloride |
Other cations |
manganese(IV) oxide Technetium(IV) oxide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Synthesis and reactions
It forms via comproportionation:[3]
- 2 Re2O7 + 3 Re → 7 ReO2
At high temperatures it undergoes disproportionation:
- 7 ReO2 → 2 Re2O7 + 3 Re
It forms perrhenates with alkaline hydrogen peroxide and oxidizing acids.[4] In molten sodium hydroxide it forms sodium rhenate:[5]
- 2 NaOH + ReO2 → Na2ReO3 + H2O
References
- Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). CRC Press. p. 484. ISBN 0-8493-0594-2. Retrieved 2008-06-05.
- Perry, Dale L.; Phillips, Sidney L. (1995). Handbook of Inorganic Compounds. San Diego: CRC Press. p. 328. ISBN 0-8493-8671-3. Retrieved 2008-06-05.
- G. Glemser "Rhenium (IV) Oxide" Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. p. 1480.
- "RHENIUM DIOXIDE - Manufacturer". Aaamolybdenum.com. Archived from the original on 2003-02-09. Retrieved 2012-08-06.
- G. Glemser "Sodium Rhenate (IV)" Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. p. 1483.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.