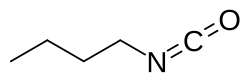
Butyl isocyanate
 | |
| Names | |
|---|---|
| IUPAC name
1-isocyanatobutane
| |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.003.512 |
| EC Number |
|
Gmelin Reference
|
662892 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C5H9NO |
| Molar mass | 99.133 g·mol−1 |
| Appearance | colourless liquid with a pungent odor |
| Density | 0.89 g/cm3 |
| Melting point | −75 °C (−103 °F; 198 K) |
| Boiling point | 115 °C (239 °F; 388 K) |
Solubility in water
|
decomposes in water |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
   
|
Signal word
|
Danger |
Hazard statements
|
H224, H225, H302, H311, H314, H317, H330, H334, H335 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P260, P262, P264, P264+P265, P270, P271, P272, P280, P284, P301+P317, P301+P330+P331, P302+P352, P302+P361+P354, P303+P361+P353, P304+P340, P305+P354+P338, P316, P317, P319, P320, P321, P330, P333+P317, P342+P316, P361+P364, P362+P364, P363, P370+P378, P403, P403+P233, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 
3
3
2 |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Butyl isocyanate is an organic chemical compound of carbon, hydrogen, nitrogen, and oxygen with the linear formula CH3(CH2)3NCO.[2] This is the butyl ester of isocyanic acid.[3]
Synthesis
Butyl isocyanate is usually produced by the reaction between n-butylamine and phosgene.[4][5]
Physical properties
Butyl isocyanate is a highly reactive chemical compound. Like isocyanic acid and other isocyanates, it is highly toxic. Butyl isocyanate polymerizes upon heating.[6]
The compound forms colourless, tear-inducing liquid with a pungent odor.[7]
Chemical properties
Propyl isocyanate decomposes in water, alcohols, and acids. Also decomposes on burning.[8] Butyl isocyanate can be anionically polymerized into poly(butyl isocyanate) when lanthanum isopropoxide is used as the initiator.[3]
Uses
The compound is used in production of other chemicals.
References
- ^ PubChem. "Butyl isocyanate". pubchem.ncbi.nlm.nih.gov. Retrieved 2026-01-20.
- ^ "Butyl isocyanate". TCI Chemicals. Retrieved 20 January 2026.
- ^ a b "B95736 Butyl isocyanate". Sigma Aldrich. Retrieved 20 January 2026.
- ^ Ashford, Robert D. (2001). Ashford's dictionary of industrial chemicals (2. ed.). London: Wavelength Publications. p. 189. ISBN 0-9522674-2-X. Retrieved 20 January 2026.
- ^ "Butylisocyanate - Preparation Method". chembk.com. Retrieved 20 January 2026.
- ^ Watts, Henry; Morley, Henry Forster; Muir, Matthew Moncrieff Pattison (1889). Watts' Dictionary of Chemistry. Longmans, Green. p. 315. Retrieved 20 January 2026.
- ^ "N-BUTYL ISOCYANATE" (PDF). NJ.gov. Retrieved 20 January 2026.
- ^ "ICSC 1642 - n-BUTYL ISOCYANATE". inchem.org. Retrieved 20 January 2026.