Ethyl isocyanate
 | |
| Names | |
|---|---|
| IUPAC name
Isocyanatoethane
| |
| Other names
NSC 89687
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.003.381 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
| UN number | 2481 (ETHYL ISOCYANATE) |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C3H5NO |
| Molar mass | 71.079 g·mol−1 |
| Appearance | colourless liquid with a pungent odor |
| Density | 0.91 g/cm3 |
| Boiling point | 59–61 °C (138–142 °F; 332–334 K) |
Solubility in water
|
Decomposes in water |
| Solubility | Miscible in ethanol, ether[1] soluble in chlorinated and aromatic hydrocarbons[1] |
| Vapor pressure | 17.5 mmHg[1] |
Refractive index (nD)
|
1.3808 at 20 °C/D[1] |
| Thermochemistry | |
Std enthalpy of
combustion (ΔcH⦵298) |
424.5 kcal (liquid)[1] |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
   
|
Signal word
|
Danger |
Hazard statements
|
H225, H301, H302, H312, H315, H319, H330, H332, H334, H335 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P260, P264, P264+P265, P270, P271, P280, P284, P301+P316, P301+P317, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P316, P317, P319, P320, P321, P330, P332+P317, P337+P317, P342+P316, P362+P364, P370+P378, P403, P403+P233, P403+P235, P405, P501 |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
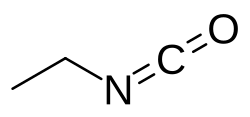
Ethyl isocyanate is an organic chemical compound of carbon, hydrogen, nitrogen and oxygen with the molecular formula C3H5NO.[2] The compound belongs to the group of isocyanates. Its structural formula is CH3—CH2—NCO.[3]
Synthesis
Ethyl isocyanate can be obtained by reacting triphosgene with ethylamine hydrochloride in xylene.
Physical properties
Ethyl isocyanate is a highly flammable, volatile, colorless liquid with a pungent odor that decomposes in water.[4]
- C3H5NO + H2O → CO2 + NH2C2H5[5]
Uses
The compound is used in the production of pharmaceuticals and pesticides.[6]
References
- ^ a b c d e f PubChem. "Ethyl isocyanate". pubchem.ncbi.nlm.nih.gov. Retrieved 2026-01-20.
- ^ Roscoe, Henry Enfield (1890). A Treatise on Chemistry: The chemistry of the hydrocarbons and their derivatives, or Organic chemistry. D. Appleton and Company. p. 420. Retrieved 19 January 2026.
- ^ "Ethyl isocyanate". Sigma Aldrich. Retrieved 18 January 2026.
- ^ "ETHYL ISOCYANATE | CAMEO Chemicals | NOAA". cameochemicals.noaa.gov. Retrieved 19 January 2026.
- ^ Bernthsen, August (1894). A text-book of organic chemistry, tr. by G. M'Gowan. p. 279. Retrieved 19 January 2026.
- ^ "Ethyl isocyanate" (PDF). NJ.gov. Retrieved 19 January 2026.