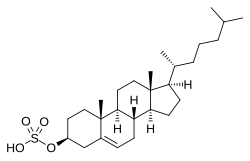
Cholesterol sulfate
Names
IUPAC name
[(3S ,8S ,9S ,10R ,13R ,14S ,17R )-10,13-Dimethyl-17-[(2R )-6-methylheptan-2-yl]-2,3,4,7,8,9,11,12,14,15,16,17-dodecahydro-1H -cyclopenta[a ]phenanthren-3-yl] hydrogen sulfate
Other names
Cholest-5-en-3β-ol sulfate
Identifiers
CAS Number
3D model (JSmol)
ChEBI
ChEMBL
ChemSpider
UNII
InChI=1S/C27H46O4S/c1-18(2)7-6-8-19(3)23-11-12-24-22-10-9-20-17-21(31-32(28,29)30)13-15-26(20,4)25(22)14-16-27(23,24)5/h9,18-19,21-25H,6-8,10-17H2,1-5H3,(H,28,29,30)/t19-,21+,22+,23-,24+,25+,26+,27-/m1/s1
Key: BHYOQNUELFTYRT-DPAQBDIFSA-N
InChI=1/C27H46O4S/c1-18(2)7-6-8-19(3)23-11-12-24-22-10-9-20-17-21(31-32(28,29)30)13-15-26(20,4)25(22)14-16-27(23,24)5/h9,18-19,21-25H,6-8,10-17H2,1-5H3,(H,28,29,30)/t19-,21+,22+,23-,24+,25+,26+,27-/m1/s1
Key: BHYOQNUELFTYRT-DPAQBDIFBL
C[C@H](CCCC(C)C)[C@H]1CC[C@@H]2[C@@]1(CC[C@H]3[C@H]2CC=C4[C@@]3(CC[C@@H](C4)OS(=O)(=O)O)C)C
Properties
Chemical formula
C 27 H 46 O 4 S
Molar mass
−1
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Cholesterol sulfate , or cholest-5-en-3β-ol sulfate , is an endogenous steroid and the C3β sulfate ester of cholesterol .[ 1] [ 2] [ 2] [ 1] [ 1] [ 2]
See also
References
Endogenous steroids
Precursors
Cholesterol 22R -Hydroxycholesterol
20α,22R -Dihydroxycholesterol
Pregnenolone 11β-Hydroxypregnenolone
17α-Hydroxypregnenolone
21-Hydroxypregnenolone
17α,21-Dihydroxypregnenolone
11β,17α,21-Trihydroxypregnenolone Corticosteroids
Glucocorticoids
Metabolites: 5α-Dihydrocortisol3α,5α-Tetrahydrocortisol Mineralocorticoids
Sex steroids
Androgens
Metabolites: 3α-Androstanediol3α-Androstanediol glucuronide
3β-Androstanediol
5β-Dihydrotestosterone
3α-Etiocholanediol
3β-Etiocholanediol
Androstanetriols
Androstenediol sulfate
Androstenetriol
Androsterone glucuronide
Androsterone sulfate
Dihydrotestosterone glucuronide
Dihydrotestosterone sulfate
Etiocholanedione
Etiocholanolone Etiocholanolone glucuronide
Epietiocholanolone
Testosterone glucuronide
Testosterone sulfate Estrogens
Metabolites: 2-Methoxyestradiol 2-Methoxyestrone
2-Methoxyestriol
4-Methoxyestriol
Estradiol disulfate
Estradiol glucuronide
Estradiol 3-glucuronide
Estradiol 3-glucuronide 17β-sulfate
Estradiol sulfate
Estradiol 17β-sulfate
Estrone glucuronide
Estrone sulfate Estriol glucuronide
Estriol sulfate
Lipoidal estradiol (e.g., estradiol stearate, estradiol palmitate) Progestogens
Metabolites: AllopregnanediolPregnanediol Pregnanediol glucuronide
Pregnanetriol
Neurosteroids Others
Mevalonate pathway
Non-mevalonate pathway
To Cholesterol From Cholesterol
22R -Hydroxycholesterol
20α,22R -Dihydroxycholesterol
See here instead. Nonhuman