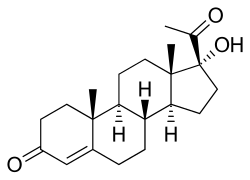
17α-Hydroxyprogesterone
Names
IUPAC name
17α-Hydroxypregn-4-ene-3,20-dione
Systematic IUPAC name
(1R ,3aS ,3bR ,9aR ,9bS ,11aS )-1-Acetyl-1-hydroxy-9a,11a-dimethyl-1,2,3,3a,3b,4,5,8,9,9a,9b,10,11,11a-tetradecahydro-7H -cyclopenta[a ]phenanthren-7-one
Other names
Hydroxyprogesterone (
INN Tooltip International Nonproprietary Name )
Identifiers
CAS Number
3D model (JSmol)
ChEBI
ChEMBL
ChemSpider
ECHA InfoCard
100.000.636
IUPHAR/BPS
KEGG
UNII
InChI=1S/C21H30O3/c1-13(22)21(24)11-8-18-16-5-4-14-12-15(23)6-9-19(14,2)17(16)7-10-20(18,21)3/h12,16-18,24H,4-11H2,1-3H3/t16-,17+,18+,19+,20+,21+/m1/s1
Key: DBPWSSGDRRHUNT-CEGNMAFCSA-N
CC(=O)[C@]1(CC[C@@H]2[C@@]1(CC[C@H]3[C@H]2CCC4=CC(=O)CC[C@]34C)C)O
Properties
Chemical formula
C21 H30 O3
Molar mass
330.46 g/mol
Melting point
219.5
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
17α-Hydroxyprogesterone (17α-OHP ), also known as 17-OH progesterone (17-OHP ),[ 1] hydroxyprogesterone (OHP ), is an endogenous progestogen steroid hormone related to progesterone .[ 2] [ 3] [ 4]
Biological activity
17α-OHP is an agonist of the progesterone receptor (PR) similarly to progesterone, albeit weakly in comparison.[ 5] [ 6] 50 >100-fold less relative to cortisol ) at the latter site, also similarly to progesterone.[ 5] [ 7] [ 8]
Relative affinities (%) of hydroxyprogesterone and related steroids
Compound
hPR-A hPR-B rbPR rbGR rbER
Progesterone 100
100
100
<1
<1
17α-Hydroxyprogesterone
1
1
3
1
<1
Hydroxyprogesterone caproate
26
30
28
4
<1
Hydroxyprogesterone acetate
38
46
115
3
?
Notes: Values are percentages (%). Reference ligands (100%) were progesterone for the PR Tooltip progesterone receptor , dexamethasone for the GR Tooltip glucocorticoid receptor , and estradiol for the ER Tooltip estrogen receptor . Sources: See template.
Biochemistry
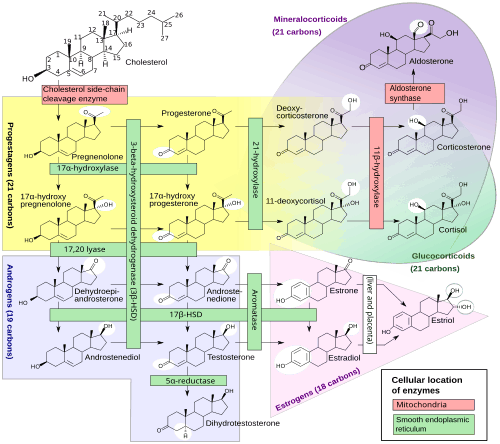
Steroidogenesis, showing 17α-OHP around the left-middle among the pregnenes.
Biosynthesis
17α-OHP is derived from progesterone via 17α-hydroxylase (encoded by CYP17A1).[ 9]
17α-OHP increases in the third trimester of pregnancy primarily due to fetal adrenal production.[ 10]
This steroid is primarily produced in the adrenal glands and to some degree in the gonads, specifically the corpus luteum of the ovary. Normal levels are 3-90 ng/dl in children, and in women, 20-100 ng/dl prior to ovulation, and 100-500 ng/dl during the luteal phase.[ 11] [ 12]
Measurement
Measurements of levels of 17α-OHP are useful in the evaluation of patients with suspected congenital adrenal hyperplasia as the typical enzymes that are defective, namely 21-hydroxylase and 11β-hydroxylase, lead to a build-up of 17α-OHP.[ 13] [ 9] [ 14]
Immunoassays like RIA (radioimmunoassay) or IRMA (immunoradiometric
assay) used to clinically determine 17α-OHP are prone to cross-reactivity with the 17α-OHP steroid precursors and their sulphated conjugates. Gas or liquid chromatography and mass spectrometry (e.g. LC-MS/MS) achieves greater specificity than immunoassays.[ 15] [ 16]
Measurement of 17α-OHP by LC-MS/MS improves newborn screening for congenital adrenal hyperplasia due to 21-hydroxylase deficiency, because 17α-OHP steroid precursors and their sulphated conjugates which are present in the first two days after birth and longer in pre-term neonates, cross-react in immunoassays with 17α-OHP, giving falsely high 17α-OHP levels.[ 15] [ 16]
Pharmacology
Pharmacokinetics
Although 17α-OHP has not been used as a medication, its pharmacokinetics have been studied and reviewed.[ 17]
Medical uses
Esters of 17α-OHP, such as hydroxyprogesterone caproate and, to a far lesser extent, hydroxyprogesterone acetate and hydroxyprogesterone heptanoate, have been used in medicine as progestins.[ 2] [ 3] [ 4]
Chemistry
17α-OHP is the parent compound of a class of progestins referred to as the 17α-hydroxyprogesterone derivatives .[ 18] [ 19] [ 20] medroxyprogesterone acetate , and megestrol acetate.[ 18] [ 19] [ 20]
Society and culture
Generic names
Hydroxyprogesterone is the generic name of 17α-OHP and its INN Tooltip International Nonproprietary Name and BAN Tooltip British Approved Name .[ 2] [ 3] [ 4]
See also
References
^ "17-hydroxyprogesterone (17OHP)" .^ a b c J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies 664– 665. ISBN 978-1-4757-2085-3 . ^ a b c I.K. Morton, Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms 978-94-011-4439-1 . ^ a b c Index Nominum 2000: International Drug Directory 978-3-88763-075-1 .^ a b Attardi BJ, Zeleznik A, Simhan H, Chiao JP, Mattison DR, Caritis SN (2007). "Comparison of progesterone and glucocorticoid receptor binding and stimulation of gene expression by progesterone, 17-alpha hydroxyprogesterone caproate, and related progestins" . Am. J. Obstet. Gynecol . 197 (6): 599.e1–7. doi:10.1016/j.ajog.2007.05.024 . PMC 2278032 18060946 . ^ Mooij CF, Parajes S, Pijnenburg-Kleizen KJ, Arlt W, Krone N, Claahsen-van der Grinten HL (April 2015). "Influence of 17-Hydroxyprogesterone, Progesterone and Sex Steroids on Mineralocorticoid Receptor Transactivation in Congenital Adrenal Hyperplasia" (PDF) . Horm Res Paediatr . 83 (6): 414– 421. doi:10.1159/000374112 . PMID 25896481 . S2CID 24727940 . ^ Pijnenburg-Kleizen KJ, Engels M, Mooij CF, Griffin A, Krone N, Span PN, van Herwaarden AE, Sweep FC, Claahsen-van der Grinten HL (2015). "Adrenal Steroid Metabolites Accumulating in Congenital Adrenal Hyperplasia lead to Transactivation of the Glucocorticoid Receptor" . Endocrinology . 156 (10): 3504– 3510. doi:10.1210/en.2015-1087 26207344 . ^ Sun K, Lei K, Chen L, Georgiou EX, Sooranna SR, Khanjani S, Brosens JJ, Bennett PR, Johnson MR (2012). "Progesterone Acts via the Nuclear Glucocorticoid Receptor to Suppress IL-1β-Induced COX-2 Expression in Human Term Myometrial Cells" . PLOS ONE . 7 (11) e50167. Bibcode:2012PLoSO...750167L . doi:10.1371/journal.pone.0050167 1932-6203 . PMC 3509141 23209664 . ^ a b Kim SM, Rhee JH (2015). "A case of 17 alpha-hydroxylase deficiency" . Clinical and Experimental Reproductive Medicine . 42 (2). The Korean Society for Reproductive Medicine: 72– 76. doi:10.5653/cerm.2015.42.2.72 . ISSN 2233-8233 . PMC 4496435 26161337 . ^ Tal R, Taylor HS (2021-03-18). "Endocrinology of Pregnancy" . MDText.com, Inc. PMID 25905197 . Retrieved 2024-06-25 . ^ Reference Values During Pregnancy ^ "normal ranges for hormone tests in women" . Archived from the original on 2020-11-08. Retrieved 2011-08-07 .^ Held PK, Bird IM, Heather NL (2020-08-23). "Newborn Screening for Congenital Adrenal Hyperplasia: Review of Factors Affecting Screening Accuracy" . International Journal of Neonatal Screening . 6 (3). MDPI AG: 67. doi:10.3390/ijns6030067 2409-515X . PMC 7569755 33117906 . ^ Check JH, Vaze MM, Epstein R, Wu CH, Quattrocchi J, Vetter B (1990). "17-Hydroxyprogesterone level as a marker for corpus luteum function in aborters versus nonaborters". International Journal of Fertility . 35 (2): 112– 115. ISSN 0020-725X . PMID 1970979 . ^ a b de Hora MR, Heather NL, Patel T, Bresnahan LG, Webster D, Hofman PL (March 2020). "Measurement of 17-Hydroxyprogesterone by LCMSMS Improves Newborn Screening for CAH Due to 21-Hydroxylase Deficiency in New Zealand" . International Journal of Neonatal Screening . 6 (1): 6. doi:10.3390/ijns6010006 7422986 33073005 . ^ a b Bialk ER, Lasarev MR, Held PK (September 2019). "Wisconsin's Screening Algorithm for the Identification of Newborns with Congenital Adrenal Hyperplasia" . International Journal of Neonatal Screening . 5 (3): 33. doi:10.3390/ijns5030033 7510207 33072992 . ^ Die Gestagene 276– 277. ISBN 978-3-642-99941-3 .^ a b Jeffrey K. Aronson (21 February 2009). Meyler's Side Effects of Endocrine and Metabolic Drugs 978-0-08-093292-7 . ^ a b Robert Alan Prentky, Ann Wolbert Burgess (31 July 2000). Forensic Management of Sexual Offenders 978-0-306-46278-8 . ^ a b H. J. Smith, Hywel Williams (1 January 1983). Introduction to the Principles of Drug Design 978-1-4831-8350-3 .
Endogenous steroids
Precursors
Cholesterol 22R -Hydroxycholesterol
20α,22R -Dihydroxycholesterol
Pregnenolone 11β-Hydroxypregnenolone
17α-Hydroxypregnenolone
21-Hydroxypregnenolone
17α,21-Dihydroxypregnenolone
11β,17α,21-Trihydroxypregnenolone Corticosteroids
Glucocorticoids
Metabolites: 5α-Dihydrocortisol3α,5α-Tetrahydrocortisol Mineralocorticoids
Sex steroids
Androgens
Metabolites: 3α-Androstanediol3α-Androstanediol glucuronide
3β-Androstanediol
5β-Dihydrotestosterone
3α-Etiocholanediol
3β-Etiocholanediol
Androstanetriols
Androstenediol sulfate
Androstenetriol
Androsterone glucuronide
Androsterone sulfate
Dihydrotestosterone glucuronide
Dihydrotestosterone sulfate
Etiocholanedione
Etiocholanolone Etiocholanolone glucuronide
Epietiocholanolone
Testosterone glucuronide
Testosterone sulfate Estrogens
Metabolites: 2-Methoxyestradiol 2-Methoxyestrone
2-Methoxyestriol
4-Methoxyestriol
Estradiol disulfate
Estradiol glucuronide
Estradiol 3-glucuronide
Estradiol 3-glucuronide 17β-sulfate
Estradiol sulfate
Estradiol 17β-sulfate
Estrone glucuronide
Estrone sulfate Estriol glucuronide
Estriol sulfate
Lipoidal estradiol (e.g., estradiol stearate, estradiol palmitate) Progestogens
Metabolites: AllopregnanediolPregnanediol Pregnanediol glucuronide
Pregnanetriol
Neurosteroids Others
Progestogens and antiprogestogens
Progestogens
PR Tooltip Progesterone receptor agonists
17α-Hydroxyprogesterone (and closely related) derivatives: 17α-Hydroxylated: Acetomepregenol (mepregenol diacetate)Algestone acetophenide (dihydroxyprogesterone acetophenide)
Anagestone acetate
Chlormadinone acetate
Chlormethenmadinone acetate
Cyproterone acetate
Delmadinone acetate
Flugestone acetate (flurogestone acetate)
Flumedroxone acetate
Hydroxyprogesterone acetate
Hydroxyprogesterone caproate
Hydroxyprogesterone heptanoate
Medroxyprogesterone acetate # Megestrol acetate
Melengestrol acetate
Methenmadinone acetate
Osaterone acetate
Pentagestrone acetate
Proligestone; 17α-Methylated: Medrogestone; Others: Haloprogesterone 19-Norprogesterone derivatives: 17α-Hydroxylated: Gestonorone caproate (gestronol hexanoate)Nomegestrol acetate
Norgestomet
Segesterone acetate (nestorone, elcometrine); 17α-Methylated: Demegestone
Promegestone Trimegestone Others: Anabolic–androgenic steroids (e.g., nandrolone and esters, trenbolone and esters, ethylestrenol , norethandrolone, others)
Antiprogestogens
SPRMs Tooltip Selective progesterone receptor modulators
Telapristone§
Ulipristal acetate PR Tooltip Progesterone receptor antagonists
# WHO-EM‡ Withdrawn from marketClinical trials:
† Phase III§ Never to phase III
See also
Progesterone receptor modulators
Androgens and antiandrogens
Estrogens and antiestrogens
List of progestogens
Biological activity
Glucocorticoid receptor modulators
GR Tooltip Glucocorticoid receptor
Agonists
Others/atypical (other expanded steroid ring systems, homosteroids, and non-pregnane steroids): CortisuzolCortivazol
Domoprednate
Naflocort
Nicocortonide
Nivacortol (nivazol)
Oxisopred
RU-26988
RU-28362 Nonsteroidal glucocorticoids: AZD-5423GSK-9027 MixedSEGRMs Tooltip Selective glucocorticoid receptor agonists )
Dagrocorat
Fosdagrocorat
Mapracorat Antagonists
7α-Hydroxy-DHEA
17α-Methylprogesterone
Aglepristone
Asoprisnil
Asoprisnil ecamate
C108297
C113176
CORT-108297
Cyproterone acetate
Dazucorilant
Exicorilant (CORT-125281)
Guggulsterone
Ketoconazole Lilopristone
LLY-2707
Metapristone (RU-42633)
Miconazole Mifepristone (RU-486) Miricorilant (CORT-118335)
Onapristone
ORG-34116
ORG-34517 (SCH-900636)
ORG-34850
Pregnenolone 16α-carbonitrile
Relacorilant (CORT-125134)
RTI 3021–012
RTI 3021–022
Telapristone
Tibolone
Toripristone
Ulipristal acetate
Zavacorilant Others
Antisense oligonucleotides: IONIS-GCCRRx (ISIS-426115)
See also
Receptor/signaling modulators
Glucocorticoids and antiglucocorticoids
Mineralocorticoid receptor modulators
List of corticosteroids
Mineralocorticoid receptor modulators
MR Tooltip Mineralocorticoid receptor
Agonists Antagonists
Steroidal: 6β-Hydroxy-7α-thiomethylspironolactone7α-Acetylthio-17α-hydroxyprogesterone
7α-Thiomethylspironolactone (SC-26519)
7α-Thioprogesterone (SC-8365)
7α-Thiospironolactone (SC-24813)
16α-Hydroxyprogesterone 17α-Hydroxyprogesterone (hydroxyprogesterone) 18-Deoxyaldosterone
18,19-Dinorprogesterone
Canrenoate potassium (potassium canrenoate)
Canrenoic acid (canrenoate)
Canrenone (canrenoate y-lactone)
Dicirenone
Dimethisterone
Drospirenone Dydrogesterone Eplerenone Gestodene Guggulsterone
Hydroxyprogesterone caproate
Medrogestone
Mespirenone
Metribolone Mexrenoate potassium
Mexrenoic acid (mexrenoate)
Mexrenone
Oxprenoic acid (oxprenoate)
Oxprenoate potassium (RU-28318)
Pregnenolone Progesterone Prorenoate potassium
Prorenoic acid (prorenoate)
Prorenone
RO-14-9012
RU-26752
SC-5233 (spirolactone)
SC-8109
SC-11927 (CS-1)
SC-19886
SC-27169
Spirorenone
Spironolactone Spiroxasone
Tibolone
Trimegestone
Vamorolone
ZK-91587
ZK-97894
See also
Receptor/signaling modulators
Mineralocorticoids and antimineralocorticoids
Glucocorticoid receptor modulators
List of corticosteroids
Progesterone receptor modulators
PR Tooltip Progesterone receptor
Agonists
Retroprogesterone derivatives: 20α-Dihydrodydrogesterone20α-Dihydrotrengestone
DU-41164
DU-41165
Dydrogesterone Retroprogesterone
Ro 6-3129
Trengestone 17α-Substituted progesterone derivatives: 6α-Methyl-17α-bromoprogesterone15β-Hydroxycyproterone acetate
16-Methylene-17α-hydroxyprogesterone acetate
17α-Bromoprogesterone
17α-Hydroxyprogesterone (hydroxyprogesterone) 17α-Methylprogesterone
Acetomepregenol (mepregenol diacetate)
Algestone
Algestone acetonide
Algestone acetophenide
Anagestone
Anagestone acetate
Bromethenmadinone
Bromethenmadinone acetate
Butagest (buterol)
Chlormadinone
Chlormadinone acetate
Chlormadinone caproate
Chlormethenmadinone
Chlormethenmadinone acetate
Cismadinone
Cismadinone acetate
Clogestone
Clogestone acetate
Clomegestone
Clomegestone acetate
Cymegesolate
Cyproterone acetate
Delmadinone
Delmadinone acetate
Edogestrone
Flugestone
Flugestone acetate
Fluorometholone Fluorometholone acetate
Flumedroxone
Flumedroxone acetate
Fluoromedroxyprogesterone acetate
Gestaclone
Gestobutanoyl
Haloprogesterone
Hydromadinone
Hydromadinone acetate
Hydroxyprogesterone acetate
Hydroxyprogesterone caproate (hydroxyprogesterone hexanoate)
Hydroxyprogesterone heptanoate (hydroxyprogesterone enanthate)
Hydroxyprogesterone heptanoate benzilic acid hydrazone
Mecigestone (pentarane B)
Medrogestone
Medroxyprogesterone Medroxyprogesterone acetate Medroxyprogesterone caproate
Megestrol Megestrol acetate
Megestrol caproate
Melengestrol
Melengestrol acetate
Methenmadinone
Methenmadinone acetate
Methenmadinone caproate
Mometasone Mometasone furoate Osaterone
Osaterone acetate
Pentagestrone
Pentagestrone acetate
Pentarane A
Proligestone
Triamcinolone acetonide 19-Norprogesterone derivatives: 17α-Methyl-19-norprogesterone18-Methylsegesterone acetate
19-Norprogesterone
Amadinone
Amadinone acetate
Demegestone
Fluoro ethyl norprogesterone
Fluoro furanyl norprogesterone
Gestadienol
Gestadienol acetate
Gestonorone acetate (gestronol acetate)
Gestonorone caproate (gestronol hexanoate)
Gestronol (gestonorone)
Nomegestrol
Nomegestrol acetate
Norgestomet
ORG-2058
Oxogestone
Oxogestone phenpropionate (xinogestone)
Promegestone Segesterone
Segesterone acetate (nestorone)
Trimegestone Testosterone derivatives: Progestins: 6,6-Difluoronorethisterone6,6-Difluoronorethisterone acetate
17α-Allyl-19-nortestosterone
Allylestrenol Altrenogest
Chloroethynylnorgestrel
Cingestol
Danazol Desogestrel Dienogest
Ethinylandrostenediol
Ethisterone Ethynerone
Etonogestrel Etynodiol
Etynodiol diacetate Gestodene Gestrinone Levonorgestrel Levonorgestrel esters (e.g., levonorgestrel butanoate)
Lynestrenol
Lynestrenol phenylpropionate
Metynodiol
Metynodiol diacetate
Norelgestromin
Norethisterone (norethindrone) Norethisterone esters (e.g., norethisterone acetate, norethisterone enanthate)
Noretynodrel Norgesterone
Norgestimate Norgestrel Norgestrienone
Norvinisterone
Oxendolone
Quingestanol
Quingestanol acetate
Tibolone
Tigestol
Tosagestin; Anabolic–androgenic steroids: 11β-Methyl-19-nortestosterone
11β-Methyl-19-nortestosterone dodecylcarbonate
19-Nor-5-androstenediol
19-Nor-5-androstenedione
19-Nordehydroepiandrosterone
Bolandiol
Bolandiol dipropionate
Bolandione
Dimethisterone
Dienedione
Dienolone
Dimethandrolone
Dimethandrolone buciclate
Dimethandrolone dodecylcarbonate
Dimethandrolone undecanoate
Dimethyldienolone
Dimethyltrienolone
Ethyldienolone
Ethylestrenol (ethylnandrol) Methyldienolone
Metribolone (R-1881) Methoxydienone (methoxygonadiene)
Mibolerone
Nandrolone Nandrolone esters (e.g., nandrolone decanoate, nandrolone phenylpropionate)
Norethandrolone
Normethandrone (methylestrenolone, normethandrolone, normethisterone)
RU-2309
Tetrahydrogestrinone
Trenbolone (trienolone)
Trenbolone esters (e.g., trenbolone acetate, trenbolone enanthate)
Trendione
Trestolone
Trestolone acetate Spirolactone derivatives: Canrenoic acidCanrenone
Drospirenone Mespirenone
Potassium canrenoate
Prorenone
SC-5233 (spirolactone)
SC-8109
Spironolactone Spirorenone Nonsteroidal: 3,8-DihydrodiligustilideLG-2527
LG-100128
Riligustilide
RWJ-26819
RWJ-49853
RWJ-60130
Tanaproget ZM-182345 Unknown: ORG-47241ORG-201745 MixedSPRMs Tooltip Selective progesterone receptor modulators )
Steroidal: Dihydroethisterone5α-Dihydrolevonorgestrel
5α-Dihydronorethisterone
Asoprisnil
Asoprisnil ecamate
Guggulsterone
J1042
LG-120838
Metapristone (RU-42633)
Mifepristone (RU-486) ORF-9371
ORF-9326
ORG-31710
ORG-33628
RMI-12936
Telapristone
Ulipristal acetate
Vilaprisan
ZK-137316 Antagonists
Steroidal: AglepristoneLilopristone
Lonaprisan
Onapristone
ORG-31710
ORG-31806
ORG-33628
RTI 3021–022
Toripristone
Zanoterone Nonsteroidal: DarolutamideLG-001447
LG-100127
LG-100128
LG-120830
LG-121046
Valproic acid ZM-150271
ZM-172406
mPR Tooltip Membrane progesterone receptor PAQR Tooltip Progestin and adipoQ receptor )
See also
Receptor/signaling modulators
Progestogens and antiprogestogens
Androgen receptor modulators
Estrogen receptor modulators
List of progestogens
Xenobiotic-sensing receptor modulators
CAR Tooltip Constitutive androstane receptor PXR Tooltip Pregnane X receptor
See also
Receptor/signaling modulators