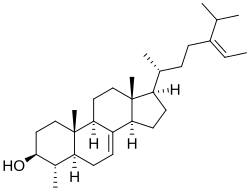
Citrostadienol
 | |
| Names | |
|---|---|
| IUPAC name
(3S,4S,5S,9R,10S,13R,14R,17R)-4,10,13-trimethyl-17-[(Z,2R)-5-propan-2-ylhept-5-en-2-yl]-2,3,4,5,6,9,11,12,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthren-3-ol
| |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
Beilstein Reference
|
3222151 |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C30H50O |
| Molar mass | 426.729 g·mol−1 |
| Appearance | powder |
| Density | 0.96 g/cm3[1] |
| Melting point | 161.6 |
Solubility in water
|
poorly soluble |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Citrostadienol is a naturally occurring phytosterol with the chemical formula C30H50O, obtained from citrus species. As a 4-desmethylsterol[2][3], it is a biosynthetic precursor in the plant sterol pathway and has attracted scientific interest for its potential biological activities, particularly its antimicrobial properties.[4] The compound also belongs to the sterol class of steroids.[5]
Structure
The сitrostadienol structure is characterized by a double bond between the C-7 and C-8 positions (a Δ⁷ bond) in the sterol nucleus, classifying it as a Δ⁷-sterol. This fact distinguishes it from more common plant sterols like β-sitosterol or stigmasterol, which are typically Δ⁵-sterols.[6]
Natural occurrence
The compound is found primarily in the Rutaceae family, especially citrus species, and in grains like oats (genus Avena). The compound is also present in the fruits of Schizandra chinensis.[7]
Synthesis
In plants, citrostadienol is synthesized via the acetate-mevalonate pathway.[8] It is an intermediate in the biosynthesis of other sterols, formed from epoxysqualene and further metabolized. Its accumulation varies among plant species and tissues.
Physical properties
The compound forms powder. Poorly soluble in water, but soluble in chloroform, dichloromethane, ethyl acetate, DMSO, acetone, etc.[9]
Uses
Ongoing scientific investigations are exploring its potential as a natural antimicrobial for diverse uses.[10] These include food preservation, where it could combat spoilage organisms to prolong product shelf life, and pharmaceuticals, especially in topical formulations for bacterial or fungal infections. Studies are also evaluating its effectiveness and safety to determine its suitability for inclusion in antimicrobial consumer products.[11][12]
References
- ^ "Citrostadienol | TargetMol". TargetMol.com. Retrieved 9 January 2026.
- ^ Sikorski, Zdzislaw Z. E.; Kolakowska, Anna (12 December 2010). Chemical and Functional Properties of Food Lipids. CRC Press. p. 101. ISBN 978-1-4200-3199-7. Retrieved 9 January 2026.
- ^ Bernfeld, Peter (22 October 2013). Biogenesis of Natural Compounds. Elsevier. p. 877. ISBN 978-1-4831-8409-8. Retrieved 9 January 2026.
- ^ Hesham, Abd El-Latif; Upadhyay, Ram Sanmukh; Sharma, Gauri Dutt; Manoharachary, Chakravarthula; Gupta, Vijai Kumar (18 June 2020). Fungal Biotechnology and Bioengineering. Springer Nature. p. 287. ISBN 978-3-030-41870-0. Retrieved 9 January 2026.
- ^ Mazur, Yehuda; Weizmann, Anna; Sondheimer, Franz (1 December 1958). "Steroids and Triterpenoids of Citrus Fruit, III, The Structure of Citrostadienol,a Natural 4Alfa-Methylstetol". Journal of the American Chemical Society. 80 (23): 6293–6296. Bibcode:1958JAChS..80.6293M. doi:10.1021/ja01556a032. ISSN 0002-7863. Retrieved 10 January 2026.
- ^ Mazur, Yehuda; Weizmann, Anna; Sondheimer, Franz (1 February 1958). "THE STRUCTURE OF CITROSTADIENOL, A NATURAL 4α-METHYLSTEROL". Journal of the American Chemical Society. 80 (4): 1007–1008. Bibcode:1958JAChS..80.1007M. doi:10.1021/ja01537a073. ISSN 0002-7863. Retrieved 9 January 2026.
- ^ Lee, Im Sean; Oh, Sei Ryang; Jung, Keun Young; Kim, Dong Sean; Kim, Jung Hee; Lee, Hyeong-Kyu (January 1997). "ANTICOMPLEMENTARY ACTIVITY AND COMPLETE 13C NMR ASSIGNMENT OF CITROSTADIENOL FROM SCHIZANDRA CHINENSIS". International Journal of Pharmacognosy. 35 (5): 358–363. doi:10.1080/09251619708951283. ISSN 0925-1618. Retrieved 9 January 2026.
- ^ Ladaniya, Milind (24 August 2022). Citrus Fruit: Biology, Technology, and Evaluation. Academic Press. p. 202. ISBN 978-0-323-99307-4. Retrieved 9 January 2026.
- ^ "Citrostadienol | CAS:474-40-8 | Steroids | High Purity | Manufacturer BioCrick". biocrick.com. Retrieved 9 January 2026.
- ^ Gong, Er Sheng; Liu, Wei; Podio, Natalia S.; You, Lijun; Zhou, Lin (27 July 2023). Dietary phytochemicals: Identification, bioactivities, and delivery strategy. Frontiers Media SA. p. 98. ISBN 978-2-8325-3005-4. Retrieved 9 January 2026.
- ^ "Citrostadienol | 474-40-8 | AAA47440 | Biosynth". biosynth.com. Retrieved 9 January 2026.
- ^ Mckenna, Dennis J.; Jones, Kenneth; Hughes, Kerry; Tyler, Virginia M. (12 November 2012). Botanical Medicines: The Desk Reference for Major Herbal Supplements, Second Edition. Routledge. p. 900. ISBN 978-1-136-39356-3. Retrieved 9 January 2026.