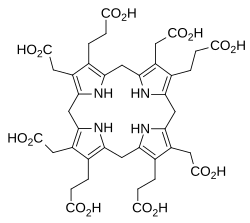
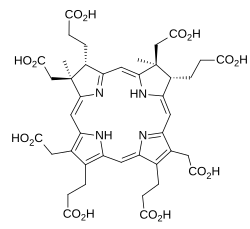
Dihydrosirohydrochlorin
 | |
| Names | |
|---|---|
| Other names
precorrin 2
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
| MeSH | 15,23-dihydrosirohydrochlorin |
PubChem CID
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C42H48N4O16 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Dihydrosirohydrochlorin is one of several naturally occurring tetrapyrrole macrocyclic metabolic intermediates in the biosynthesis of vitamin B12 (cobalamin). Its oxidised form, sirohydrochlorin, is precursor to sirohaem, the iron-containing prosthetic group in sulfite reductase enzymes. Further biosynthetic transformations convert sirohydrochlorin to cofactor F430 for an enzyme which catalyzes the release of methane in the final step of methanogenesis.[1][2]
Biosynthesis
Dihydrosirohydrochlorin is derived from a tetrapyrrolic structural framework created by the enzymes deaminase and cosynthetase which transform aminolevulinic acid via porphobilinogen and hydroxymethylbilane to uroporphyrinogen III. The latter is the first macrocyclic intermediate common to haem, chlorophyll, sirohaem and vitamin B12. Uroporphyrinogen III is subsequently transformed by the enzyme uroporphyrinogen-III C-methyltransferase which adds two methyl groups to form dihydrosirohydrochlorin.[1]
The first methylation reaction converts uroporphyrinogen III into precorrin-1 and the second forms dihydrosirohydrochlorin (precorrin-2). In both cases the methyl group comes from the cofactor, S-adenosyl methionine (SAM), which gives S-adenosyl-L-homocysteine (SAH).[3]
Biochemical reactions
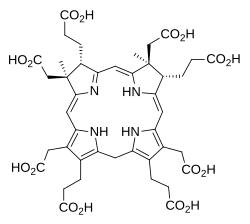
The next step in cobalamin biosynthesis introduces a third methyl group when the enzyme precorrin-2 C20-methyltransferase acts on precorrin-2:

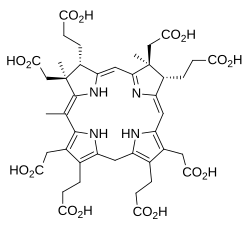
This forms precorrin-3A by methyl group insertion from SAM.[1][4]
Dihydrosirohydrochlorin is also an intermediate in the biosynthetic pathway towards siroheme and cofactor F430 via sirohydrochlorin.[5] It is oxidised by precorrin-2 dehydrogenase
The enzyme uses nicotinamide adenine dinucleotide (NAD+) as its cofactor.[6][7]
References
- ^ a b c Battersby, Alan R. (2000). "Tetrapyrroles: The pigments of life: A Millennium review". Natural Product Reports. 17 (6): 507–526. doi:10.1039/B002635M. PMID 11152419.
- ^ Mucha, Helmut; Keller, Eberhard; Weber, Hans; Lingens, Franz; Trösch, Walter (1985-10-07). "Sirohydrochlorin, a precursor of factor F430 biosynthesis in Methanobacterium thermoautotrophicum". FEBS Letters. 190 (1): 169–171. doi:10.1016/0014-5793(85)80451-8.
- ^ Enzyme 2.1.1.107 at KEGG Pathway Database.
- ^ Debussche L, Thibaut D, Cameron B, Crouzet J, Blanche F (1993). "Biosynthesis of the corrin macrocycle of coenzyme B12 in Pseudomonas denitrificans". J. Bacteriol. 175 (22): 7430–40. doi:10.1128/jb.175.22.7430-7440.1993. PMC 206888. PMID 8226690.
- ^ Mucha, Helmut; Keller, Eberhard; Weber, Hans; Lingens, Franz; Trösch, Walter (1985-10-07). "Sirohydrochlorin, a precursor of factor F430 biosynthesis in Methanobacterium thermoautotrophicum". FEBS Letters. 190 (1): 169–171. doi:10.1016/0014-5793(85)80451-8.
- ^ Warren MJ; Raux, E; Brindley, AA; Leech, HK; Wilson, KS; Hill, CP; Warren, MJ (2002). "The structure of Saccharomyces cerevisiae Met8p, a bifunctional dehydrogenase and ferrochelatase". EMBO J. 21 (9): 2068–75. doi:10.1093/emboj/21.9.2068. PMC 125995. PMID 11980703.
- ^ Warren MJ, Raux E, Schubert HL, Escalante-Semerena JC (2002). "The biosynthesis of adenosylcobalamin (vitamin B12)". Natural Product Reports. 19 (4): 390–412. doi:10.1039/b108967f. PMID 12195810.