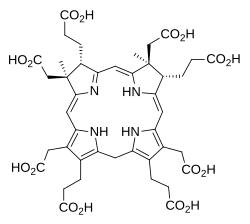
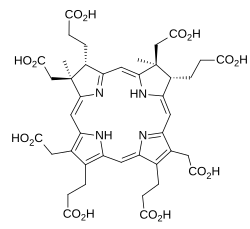
Sirohydrochlorin
 | |
| Identifiers | |
|---|---|
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C42H46N4O16 |
| Appearance | yellow solid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Sirohydrochlorin is a tetrapyrrole macrocyclic metabolic intermediate in the biosynthesis of sirohaem, the iron-containing prosthetic group in sulfite reductase enzymes. It is also the biosynthetic precursor to cofactor F430, an enzyme which catalyzes the release of methane in the final step of methanogenesis.[1]
Structure
Sirohydrochlorin was first isolated in the early 1970s when it was shown to be the metal-free form of the prosthetic group in the ferredoxin-nitrite reductase from spinach.[2] Its chemical identity was established by spectroscopy and by total synthesis.[3][4]
Biosynthesis
Sirohydrochlorin is derived from a tetrapyrrolic structural framework created by the enzymes deaminase and cosynthetase which transform aminolevulinic acid via porphobilinogen and hydroxymethylbilane to uroporphyrinogen III. The latter is the first macrocyclic intermediate common to haem, chlorophyll, sirohaem and vitamin B12. Uroporphyrinogen III is subsequently transformed by the addition of two methyl groups to form dihydrosirohydrochlorin and this is oxidised by precorrin-2 dehydrogenase to give sirohydrochlorin.[5]
The enzyme uses oxidised nicotinamide adenine dinucleotide (NAD+) as its cofactor.[6][7]
Biochemical transformations
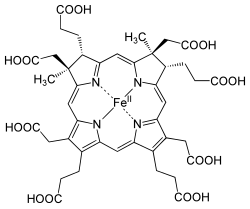
The enzyme sirohydrochlorin ferrochelatase catalyzes insertion of iron into sirohydrochlorin to form siroheme.[8][9][10]
Sirohydrochlorin cobaltochelatase inserts cobalt instead of iron.[7]
References
- ^ Mucha, Helmut; Keller, Eberhard; Weber, Hans; Lingens, Franz; Trösch, Walter (1985-10-07). "Sirohydrochlorin, a precursor of factor F430 biosynthesis in Methanobacterium thermoautotrophicum". FEBS Letters. 190 (1): 169–171. doi:10.1016/0014-5793(85)80451-8.
- ^ Murphy, M. J.; Siegel, L. M.; Tove, S. R.; Kamin, H. (1974). "Siroheme: A New Prosthetic Group Participating in Six-Electron Reduction Reactions Catalyzed by Both Sulfite and Nitrite Reductases". Proceedings of the National Academy of Sciences. 71 (3): 612–616. Bibcode:1974PNAS...71..612M. doi:10.1073/pnas.71.3.612. PMC 388061. PMID 4595566.
- ^ Scott, A. Ian; Irwin, Anthony J.; Siegel, Lewis M.; Shoolery, J. N. (1978). "Sirohydrochlorin. Prosthetic group of sulfite and nitrite reductases and its role in the biosynthesis of vitamin B12". Journal of the American Chemical Society. 100 (25): 7987–7994. doi:10.1021/ja00493a031.
- ^ Block, Michael H.; Zimmerman, Steven C.; Henderson, Graeme B.; Turner, Simon P. D.; Westwood, Steven W.; Leeper, Finian J.; Battersby, Alan R. (1985). "Syntheses relevant to vitamin B12 biosynthesis: Synthesis of sirohydrochlorin and of its octamethyl ester". Journal of the Chemical Society, Chemical Communications (16): 1061. doi:10.1039/C39850001061.
- ^ Battersby, Alan R. (2000). "Tetrapyrroles: The pigments of life: A Millennium review". Natural Product Reports. 17 (6): 507–526. doi:10.1039/B002635M. PMID 11152419.
- ^ Enzyme 1.3.1.76 at KEGG Pathway Database.
- ^ a b Warren MJ, Raux E, Schubert HL, Escalante-Semerena JC (2002). "The biosynthesis of adenosylcobalamin (vitamin B12)". Nat. Prod. Rep. 19 (4): 390–412. doi:10.1039/b108967f. PMID 12195810.
- ^ Saha, Kaushik; Webb, Michael E.; Rigby, Stephen E. J.; Leech, Helen K.; Warren, Martin J.; Smith, Alison G. (2012). "Characterization of the evolutionarily conserved iron–sulfur cluster of sirohydrochlorin ferrochelatase from Arabidopsis thaliana". Biochemical Journal. 444 (2): 227–237. doi:10.1042/BJ20111993. ISSN 0264-6021. PMID 22414210.

- ^ Warren MJ; Raux, E; Brindley, AA; Leech, HK; Wilson, KS; Hill, CP; Warren, MJ (2002). "The structure of Saccharomyces cerevisiae Met8p, a bifunctional dehydrogenase and ferrochelatase". EMBO J. 21 (9): 2068–75. doi:10.1093/emboj/21.9.2068. PMC 125995. PMID 11980703.
- ^ Enzyme 4.99.1.4 at KEGG Pathway Database.