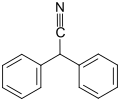
Diphenylacetonitrile
 | |
| Names | |
|---|---|
| Other names
2,2-Diphenylacetonitrile, Dipan, Diphenatrile, Benzhydrylcyanide.
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.511 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C14H11N |
| Molar mass | 193.24 g/mol |
| Density | 1.1061g/cm3 (estimated) |
| Melting point | 71–73 °C (160–163 °F; 344–346 K) |
| Boiling point | 181 °C (358 °F; 454 K) @ 12 mmHg |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
 
|
Signal word
|
Danger |
Hazard statements
|
H301, H315, H319, H335 |
Precautionary statements
|
P261, P264, P264+P265, P270, P271, P280, P301+P316, P302+P352, P304+P340, P305+P351+P338, P319, P321, P330, P332+P317, P337+P317, P362+P364, P403+P233, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Diphenylacetonitrile is an organic compound with the formula (C6H5)2CHCN. A colorless solid, it is widely used in the pharmaceutical industry.
Synthesis
In Hoch's original procedure, phenylacetonitrile was halogenated with molecular bromine to give bromobenzyl cyanide (BBC). This species was then reacted with benzene in the presence of aluminium trichloride to afford diphenylacetonitrile.[2] This procedure has been optimized over the years.[3][4]
An alternative route involves the reaction of bromodiphenylmethane with sodium cyanide under phase transfer conditions:[5]
- (C6H5)2CHBr + NaCN → (C6H5)2CHCN + NaBr
Applications
- Opioid analgesics: Bezitramide, Dipipanone, Dipyanone, Isomethadol, LAAM, Methadone,[6][7] Noracymethadol, Normethadone, Norpipanone, Piritramide, R-4066, R 4837 [59708-47-3], R-5260 [1109-69-9] (25ii):[8]
- Psychostimulants: 2-Benzhydrylpiperazine [28217-87-0],[9][10] Desoxypipradrol, DPA-1 [3139-55-7], β-Phenylmethamphetamine, Desoxyhexapradol.
- Antidepressants: McN-4187,[11] McN 4612-z[12][13] (Lead compound/antecedent to JNJ-7925476, McN 5707 & McN5652, etc. SAR with tametraline) & deschloro-clemeprol (although the literature procedure for 1,1-diphenylacetone is different to this).
- Antipsychotics: McN-4171[14][15] This agent is an analog of butaclamol. McN-4612-Y is also described in the attached article.[14] This is similar to McN 4612-z, which was described above except it is the opposite optical antipode. It is also possible to make PC12515636 from the primary amine using a double Michael reaction followed by Dieckman condensation, which is the standard method of 4-piperidone synthesis.
- Antispasmodics: Aminopentamide,[16] Buzepide,[17] Fenpiverinium, Fenpipramide
- Urinary incontinence: Darifenacin, Imidafenacin
- Anti-diarrhoeals: Difenoxin, Diphenoxylate, Nufenoxole
- Choleretic and spasmolytic agent: Diisopromine
- Analeptic (respiratory stimulant): Doxapram
- Antiarrhythmic: Isopropanamide[18][19]
- Cytochromes P450 inhibitor: Proadifen
- 2,2-DEP [36794-51-1][20] (Hamilton Morris's diphenidine positional isomer drug).
See also
- Hoch also discovered the Hoch-Campbell ethylenimine synthesis.
- Diphenylacetic acid
References
- ^ "Diphenylacetonitrile". PubChem. U.S. National Library of Medicine.
- ^ Hoch J (1933). "General method for the preparation of di- and triarylacetonitriles". Compt. Rend. 197: 770–772.
- ^ Robb CM, Schultz EM (1948). Arnold RT, Britain JW (eds.). "Diphenylacetonitrile". Organic Syntheses. 28: 55. doi:10.15227/orgsyn.028.0055.
- ^ Ginsburg D, Baizer MM (June 1949). "Diphenylacetonitrile". Journal of the American Chemical Society. 71 (6): 2254. Bibcode:1949JAChS..71Q2254G. doi:10.1021/ja01174a512.
- ^ Kasarekar RB, Sawant SB (January 1987). "Preparation of diphenyl acetonitrile by phase transfer catalysis". Journal of Chemical Technology & Biotechnology. 38 (3): 201–208. Bibcode:1987JCTB...38..201K. doi:10.1002/jctb.280380306.
- ^ https://bbgate.com/threads/synthesis-of-methadone.193/
- ^ Golzadeh, R., Mahkam, M., Rezaii, E., Nazmi Miardan, L. (22 December 2021). "Green synthesis of methadone in eutectic solvent". Main Group Chemistry. 20 (4): 463–474. doi:10.3233/MGC-210058.
- ^ Leysen, J., Tollenaere, J. P., Koch, M. H. J., Laduron, P. (June 1977). "Differentiation of opiate and neuroleptic receptor binding in rat brain". European Journal of Pharmacology. 43 (3): 253–267. doi:10.1016/0014-2999(77)90025-5. PMID 194781.
- ^ Jongh David Karel De, Akkerman Antonie Marie, Kofman Hendrik,& Vries George De, US2997473 (1961 to Nederlandsche Combinatie voor Chemische Industrie NV).
- ^ Karel De Jongh David, et al. GB893391 (1962 to Nederlandsche Combinatie voor Chemische Industrie NV).
- ^ Maryanoff BE, Nortey SO, Gardocki JF (August 1984). "Structure-activity studies on antidepressant 2,2-diarylethylamines". Journal of Medicinal Chemistry. 27 (8): 1067–1071. doi:10.1021/jm00374a022. PMID 6747990.
- ^ Nielsen, J., Duda, N., Mokler, D., Moore, K. (February 1984). "Self-administration of central stimulants by rats: A comparison of the effects of d-amphetamine, methylphenidate and McNeil 4612". Pharmacology Biochemistry and Behavior. 20 (2): 227–232. doi:10.1016/0091-3057(84)90247-8. ISSN 0091-3057.
- ^ Maryanoff, B. E., Shank, R. P., Gardocki, J. F. (1986). "McN-5707 and McN-5652-Z". Drugs of the Future. 11 (1): 18. doi:10.1358/dof.1986.011.01.51629. ISSN 0377-8282.
- ^ a b Chrzanowski, F. A., McGrogan, B. A., Maryanoff, B. E. (March 1985). "The pKa of butaclamol and the mode of butaclamol binding to central dopamine receptors". Journal of Medicinal Chemistry. 28 (3): 399–400. doi:10.1021/jm00381a022.
- ^ Kukla, M. J., Bloss, J. L., Brougham, L. R. (April 1979). "Use of the butaclamol template in a search for antipsychotic agents with lessened side effects". Journal of Medicinal Chemistry. 22 (4): 401–406. doi:10.1021/jm00190a009.
- ^ Ginsburg D, Baizer MM (August 1957). "Antispasmodics. X. α,α-Diphenyl-γ-amino Amides 1". Journal of the American Chemical Society. 79 (16): 4451–4457. Bibcode:1957JAChS..79.4451M. doi:10.1021/ja01573a056.
- ^ Janssen PA, Demoen PJ, Jageneau AH, Loomans JL (April 1959). "The influence of heterocyclic ring size of tertiary 2, 2-diphenyl-4-amino-butyramides on parasympatholytic activity". Journal of Medicinal and Pharmaceutical Chemistry. 1 (2): 187–194. doi:10.1021/jm50003a007. PMID 13665290.
- ^ JANSSEN P, ZIVKOVIC D, DEMOEN P, DE JONGH DK, VAN PROOSDIJ-HARTZEMA EG (1955). "Substituted phenylpropylamines. I. Synthesis and physicochemical properties of basic butyronitriles and butyramides". Archives Internationales de Pharmacodynamie et de Therapie. 103: 82–99. PMID 13259723.
- ^ Janssen Paul Adriaan Jan & Jongh David Karel De, US2884436 (1959 to Nederlandsche Combinatie voor Chemische Industrie NV).
- ^ Wallach, J., Kavanagh, P. V., McLaughlin, G., Morris, N., Power, J. D., Elliott, S. P., et al. (May 2015). "Preparation and characterization of the 'research chemical' diphenidine, its pyrrolidine analogue, and their 2,2‐diphenylethyl isomers". Drug Testing and Analysis. 7 (5): 358–367. doi:10.1002/dta.1689.