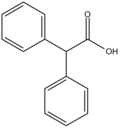
Diphenylacetic acid
 | |
| Names | |
|---|---|
| Other names
2,2-diphenylacetic acid
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.003.805 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C14H12O2 |
| Molar mass | 212.24 g/mol |
| Density | g/cm3 |
| Melting point | 147–149 °C (297–300 °F; 420–422 K) |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H302, H311, H315, H319, H331, H332, H335, H400 |
Precautionary statements
|
P261, P262, P264, P264+P265, P270, P271, P273, P280, P301+P317, P302+P352, P304+P340, P305+P351+P338, P316, P317, P319, P321, P330, P332+P317, P337+P317, P361+P364, P362+P364, P391, P403+P233, P405, P501 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Diphenylacetic acid is an organic chemical with applications in drug synthesis.[1][2][3]
Applications
- AB06-117 (cmp #23)[4]
- Adiphenine
- Arpenal
- Asimadoline
- 2,2-DEP [36794-51-1][5]
- Desmethylmoramide
- Diphenamide [957-51-7] Herbicide
- Dioxaphetyl butyrate
- Fluperamide
- Loperamide
- PD-85639
- Moramide
- Pifenate[6]
- Tropacine
- BIBO-3304 [191868-13-0]
- BIBP-3226
- PC4225440[7]
- 4-Tert-butyl-1-(2,2-diphenylethyl)-4-piperidinol (Butaclamol analog)[8]
Synthesis
- From hydroiodic acid reduction of benzilic acid.[9][10]
- Alternative procedure:[11]
- From glyoxylic acid and benzene:[15]
See also
References
- ^ Jena, A. (January 1870). "Ueber die Benzilsäure oder Diphenylglycolsäure". Justus Liebigs Annalen der Chemie. 155 (1): 77–89. doi:10.1002/jlac.18701550107.
- ^ Symons, R., Zincke, Th. (July 1873). "Ueber Diphenylessigsäure und Benzilsäure". Berichte der Deutschen Chemischen Gesellschaft. 6 (2): 1188–1191. doi:10.1002/cber.187300602102.
- ^ Symons, R., Zincke, Th. (January 1874). "Ueber Benzilsäure und Diphenylessigsäure". Justus Liebigs Annalen der Chemie. 171 (1): 117–134. doi:10.1002/jlac.18741710110.
- ^ Bonifazi, Alessandro; Battiti, Francisco O.; Sanchez, Julie; Zaidi, Saheem A.; Bow, Eric; Makarova, Mariia; Cao, Jianjing; Shaik, Anver Basha; Sulima, Agnieszka; Rice, Kenner C.; Katritch, Vsevolod; Canals, Meritxell; Lane, J. Robert; Newman, Amy Hauck (2021). "Novel Dual-Target μ-Opioid Receptor and Dopamine D3 Receptor Ligands as Potential Nonaddictive Pharmacotherapeutics for Pain Management". Journal of Medicinal Chemistry. 64 (11): 7778–7808. doi:10.1021/acs.jmedchem.1c00611. PMC 9308496. PMID 34011153.
- ^ Wallach, J., Kavanagh, P. V., McLaughlin, G., Morris, N., Power, J. D., Elliott, S. P., Mercier, M. S., Lodge, D., Morris, H., Dempster, N. M., Brandt, S. D. (May 2015). "Preparation and characterization of the 'research chemical' diphenidine, its pyrrolidine analogue, and their 2,2‐diphenylethyl isomers". Drug Testing and Analysis. 7 (5): 358–367. doi:10.1002/dta.1689.
- ^ GB granted 939019, Temple, Richard William; Wiggins, Leslie Frederick, assigned to Aspro Nicholas Ltd
- ^ Urbani, P., Cascio, M. G., Ramunno, A., Bisogno, T., Saturnino, C., Marzo, V. D. (August 2008). "Novel sterically hindered cannabinoid CB1 receptor ligands". Bioorganic & Medicinal Chemistry. 16 (15): 7510–7515. doi:10.1016/j.bmc.2008.06.001. hdl:11563/122572.
- ^ Kukla, M. J., Bloss, J. L., Brougham, L. R. (April 1979). "Use of the butaclamol template in a search for antipsychotic agents with lessened side effects". Journal of Medicinal Chemistry. 22 (4): 401–406. doi:10.1021/jm00190a009.
- ^ Demselben (January 1893). "Beiträge zur Kenntniss der Diphenylessigsäure". Justus Liebigs Annalen der Chemie. 275 (1): 83–89. doi:10.1002/jlac.18932750107.
- ^ "Diphenylacetic Acid". Organic Syntheses. 3: 45. 1923. doi:10.15227/orgsyn.003.0045.
- ^ Strazzolini, P., Giumanini, A. G., Verardo, G. (December 1987). "Diphenylacetic Acid from Benzilic Acid: An Ecologically and Economically Improved Procedure". Synthetic Communications. 17 (16): 1919–1928. doi:10.1080/00397918708057804.
- ^ Fritsch, P., Feldmann, F. (January 1899). "Synthese aromatisch disubstituirter Essigsäuren mittelst Chloral". Justus Liebigs Annalen der Chemie. 306 (1–2): 72–86. doi:10.1002/jlac.18993060106.
- ^ Kesslin, George, U.S. patent 2,756,254 (1952 to Kay-Fries Chemicals Inc).
- ^ Herbert R Elkinton, Kesslin George, & Nicholl Leonard, U.S. patent 2,517,856 (1950 to Kay-Fries Chemicals Inc).
- ^ Prakash, G. K. S., Paknia, F., Kulkarni, A., Narayanan, A., Wang, F., Rasul, G., Mathew, T., Olah, G. A. (March 2015). "Taming of superacids: PVP-triflic acid as an effective solid triflic acid equivalent for Friedel–Crafts hydroxyalkylation and acylation". Journal of Fluorine Chemistry. 171: 102–112. Bibcode:2015JFluC.171..102P. doi:10.1016/j.jfluchem.2014.08.020.