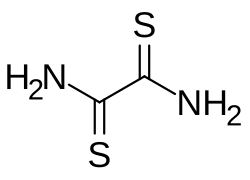
Dithiooxamide
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Ethanedithioamide | |
| Other names
dithiooxamide, rubeanic acid
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.001.095 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C2H4N2S2 |
| Molar mass | 120.19 g·mol−1 |
| Density | 1.523 g/cm3 |
| Melting point | 170 °C (338 °F; 443 K) decomp |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H302, H315, H319, H335 |
Precautionary statements
|
P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, P501 |
| Safety data sheet (SDS) | External MSDS |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Dithiooxamide, also known as rubeanic acid, is an organic compound. It is the sulfur analog of oxamide. It acts as a chelating agent, e.g. in the detection or determination of copper.[2][3] It has also been used as a building block in the synthesis of cyclen.[4]
Materials chemistry
Rubeanic acid has received much attention as a precursor to photonic or electroactive materials. It is a precursor to inorganic C-S-N rings.[5] It condenses with acid chlorides to thiazoles.[6] It forms coordination polymers.[7]
References
- ^ GHS: PubChem
- ^ "Stainsfile - Howell's rubeanic acid for copper". stainsfile.info. Archived from the original on 2006-02-13.
- ^ "Strengthen science. Advance justice".
- ^ David P. Reed and Gary R. Weisman (2004). "1,4,7,10-Tetraazacyclododecane". Organic Syntheses; Collected Volumes, vol. 10, p. 667.
- ^ McManus, Gordon D.; Rawson, Jeremy M.; Feeder, Neil; Van Duijn, Joost; McInnes, Eric J. L.; Novoa, Juan J.; Burriel, Ramon; Palacio, Fernando; Oliete, Patricia (2001). "Synthesis, crystal structures, electronic structure and magnetic behaviour of the trithiatriazapentalenyl radical, C2S3N3". Journal of Materials Chemistry. 11 (8): 1992–2003. doi:10.1039/b103303b.
- ^ Sayresmith, Nickolas A.; Saminathan, Anand; Sailer, Joshua K.; Patberg, Shannon M.; Sandor, Kristin; Krishnan, Yamuna; Walter, Michael G. (2019). "Photostable Voltage-Sensitive Dyes Based on Simple, Solvatofluorochromic, Asymmetric Thiazolothiazoles". Journal of the American Chemical Society. 141 (47): 18780–18790. doi:10.1021/jacs.9b08959. PMID 31660737.
- ^ Kanaizuka, Katsuhiko; Haruki, Rie; Sakata, Osami; Yoshimoto, Mamoru; Akita, Yasuyuki; Kitagawa, Hiroshi (2008). "Construction of Highly Oriented Crystalline Surface Coordination Polymers Composed of Copper Dithiooxamide Complexes". Journal of the American Chemical Society. 130 (47): 15778–15779. doi:10.1021/ja806799t. PMID 18983147.