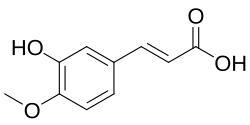
Isoferulic acid
Names
IUPAC name
(E )-3-(3-Hydroxy-4-methoxyphenyl)prop-2-enoic acid
Other names
Hesperetic acid
Identifiers
CAS Number
3D model (JSmol)
ChEBI
ChemSpider
ECHA InfoCard
100.007.889
UNII
InChI=1S/C10H10O4/c1-14-9-4-2-7(6-8(9)11)3-5-10(12)13/h2-6,11H,1H3,(H,12,13)/b5-3+
Key: QURCVMIEKCOAJU-HWKANZROSA-N
COC1=C(C=C(C=C1)C=CC(=O)O)O
Properties
Chemical formula
C 10 H 10 O 4
Molar mass
−1
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Isoferulic acid is a hydroxycinnamic acid, a type of organic compound. It is an isomer of ferulic acid .
Occurrence in nature
Isoferulic acid can be found, amongst other compounds, in Lobelia chinensis .[ 1]
In food
Ferulic acid is found in pineapple flesh.[ 2]
References
^ Chen JX, Huang SH, Wang Y, Shao M, Ye WC (2010). "Studies on the chemical constituents from Lobelia chinensis ". Zhong Yao Cai . 33 (11): 1721– 4. PMID 21434431 . ^ Edwige Sopie Yapo, Hilaire Tanoh Kouakou, Laurent kouakou kouakou, Justin Yatty Kouadio, Patrice Kouamé andjean-Michel Mérillon (2011). "Phenolic profiles of pineapple fruits (Ananas comosus L. Merrill) Influence of the origin of suckers". Australian Journal of Basic and Applied Sciences . 5 (6): 1372– 1378. {{cite journal}}: CS1 maint: multiple names: authors list (link)
Types of hydroxycinnamic acids
Aglycones
Precursor Monohydroxycinnamic acids Dihydroxycinnamic acids
Caffeic acid (3,4-dihydroxycinnamic acid)Umbellic acid (2,4-dihydroxycinnamic acid)
2,3-Dihydroxycinnamic acid
2,5-Dihydroxycinnamic acid
3,5-Dihydroxycinnamic acid Trihydroxycinnamic acids
2,4,5-Trihydroxycinnamic acid
3,4,5-Trihydroxycinnamic acid O -methylated formsothers
Esters
glycoside-likes
Esters of
esters ofquinic acid
Chlorogenic acid (3-caffeoylquinic acid)Cryptochlorogenic acid (4-O -caffeoylquinic acid)
Neochlorogenic acid (5-O -Caffeoylquinic acid)
Cynarine (1,5-dicaffeoylquinic acid)
3,4-dicaffeoylquinic acid
3,5-dicaffeoylquinic acid esters ofshikimic acid
Dactylifric acid (3-O -caffeoylshikimic acid)
Glycosides
Ferulic acid glucoside
p -Coumaric acid glucoside1-Sinapoyl-D -glucose
Tartaric acid esters
Caftaric acid
Chicoric acid (dicaffeoyltartaric acid)
Coutaric acid
Fertaric acid
Grape reaction product (caftaric acid conjugated with glutathione) Other esters Caffeoyl phenylethanoid
Echinacoside
Calceolarioside A, B, C, F
Chiritoside A, B, C
Cistanoside A, B, C, D, E, F, G, H
Conandroside
Myconoside
Pauoifloside
Plantainoside A
Plantamajoside
Tubuloside B
Verbascoside (Isoverbascoside, 2′-Acetylverbascoside)
Oligomeric forms
Dimers
Diferulic acids (DiFA) : 5,5′-Diferulic acid, 8-O -4′-Diferulic acid, 8,5′-Diferulic acid, 8,5′-DiFA (DC), 8,5′-DiFA (BF), 8,8′-Diferulic acid Trimers
Triferulic acids : 5-5′,8′-O -4″-Triferulic acid Tetramers
Conjugates withcoenzyme A (CoA)