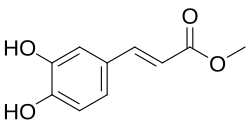
Methyl caffeate
Chemical structure of methyl caffeate
Names
Preferred IUPAC name
Methyl (2E )-3-(3,4-dihydroxyphenyl)prop-2-enoate
Other names
Caffeic acid
methyl ester
methylcaffeate
Methyl 3,4-dihydroxycinnamate
Identifiers
CAS Number
3D model (JSmol)
ChemSpider
ECHA InfoCard
100.236.052
UNII
InChI=1S/C10H10O4/c1-14-10(13)5-3-7-2-4-8(11)9(12)6-7/h2-6,11-12H,1H3/b5-3+
Key: OCNYGKNIVPVPPX-HWKANZROSA-N
InChI=1/C10H10O4/c1-14-10(13)5-3-7-2-4-8(11)9(12)6-7/h2-6,11-12H,1H3/b5-3+
Key: OCNYGKNIVPVPPX-HWKANZROBE
COC(=O)/C=C/C1=CC(=C(C=C1)O)O
Properties
Chemical formula
C 10 H 10 O 4
Molar mass
−1
Related compounds
Related compounds
Caffeic acid , Ethyl caffeate
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Methyl caffeate is an ester of caffeic acid , a naturally occurring phenolic compound. It is an α-glucosidase inhibitor.[ 1]
Natural occurrences
Methyl caffeate can be found in the fruit of Solanum torvum .[ 2]
Health effect
Methyl caffeate shows an antidiabetic effect in streptozotocin -induced diabetic rats.[ 2]
References
^ Takahashi, Keisuke; Yoshioka, Yasuyuki; Kato, Eisuke; Katsuki, Shigeki; Iida, Osamu; Hosokawa, Keizo; Kawabata, Jun (2014). "Methyl Caffeate as an α-Glucosidase Inhibitor from Solanum torvum Fruits and the Activity of Related Compounds" . Bioscience, Biotechnology, and Biochemistry . 74 (4): 741– 745. doi:10.1271/bbb.90789 2115/53430 20378981 . S2CID 23067847 . ^ a b Gandhi, Gopalsamy Rajiv; Ignacimuthu, Savarimuthu; Paulraj, Michael Gabriel; Sasikumar, Ponnusamy (2011). "Antihyperglycemic activity and antidiabetic effect of methyl caffeate isolated from Solanum torvum Swartz. Fruit in streptozotocin induced diabetic rats". European Journal of Pharmacology . 670 (2– 3): 623– 631. doi:10.1016/j.ejphar.2011.09.159 . PMID 21963451 .
Types of hydroxycinnamic acids
Aglycones
Precursor Monohydroxycinnamic acids Dihydroxycinnamic acids
Caffeic acid (3,4-dihydroxycinnamic acid)Umbellic acid (2,4-dihydroxycinnamic acid)
2,3-Dihydroxycinnamic acid
2,5-Dihydroxycinnamic acid
3,5-Dihydroxycinnamic acid Trihydroxycinnamic acids
2,4,5-Trihydroxycinnamic acid
3,4,5-Trihydroxycinnamic acid O -methylated formsothers
Esters
glycoside-likes
Esters of
esters ofquinic acid
Chlorogenic acid (3-caffeoylquinic acid)Cryptochlorogenic acid (4-O -caffeoylquinic acid)
Neochlorogenic acid (5-O -Caffeoylquinic acid)
Cynarine (1,5-dicaffeoylquinic acid)
3,4-dicaffeoylquinic acid
3,5-dicaffeoylquinic acid esters ofshikimic acid
Dactylifric acid (3-O -caffeoylshikimic acid)
Glycosides
Ferulic acid glucoside
p -Coumaric acid glucoside1-Sinapoyl-D -glucose
Tartaric acid esters
Caftaric acid
Chicoric acid (dicaffeoyltartaric acid)
Coutaric acid
Fertaric acid
Grape reaction product (caftaric acid conjugated with glutathione) Other esters Caffeoyl phenylethanoid
Echinacoside
Calceolarioside A, B, C, F
Chiritoside A, B, C
Cistanoside A, B, C, D, E, F, G, H
Conandroside
Myconoside
Pauoifloside
Plantainoside A
Plantamajoside
Tubuloside B
Verbascoside (Isoverbascoside, 2′-Acetylverbascoside)
Oligomeric forms
Dimers
Diferulic acids (DiFA) : 5,5′-Diferulic acid, 8-O -4′-Diferulic acid, 8,5′-Diferulic acid, 8,5′-DiFA (DC), 8,5′-DiFA (BF), 8,8′-Diferulic acid Trimers
Triferulic acids : 5-5′,8′-O -4″-Triferulic acid Tetramers
Conjugates withcoenzyme A (CoA)