Josamycin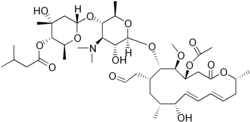 |
 |
|
| AHFS/Drugs.com | International Drug Names |
|---|
| ATC code | |
|---|
|
(2S,3S,4R,6S)-6-{[(2R,3S,4R,5R,6S)-6-{[(4R,5S,6S,7R,9R,10R,11E,13E,16R)-4-Acetoxy-10-hydroxy-5-methoxy-9,16-dimethyl-2-oxo-7-(2-oxoethyl)oxacyclohexadeca-11,13-dien-6-yl]oxy}-4-(dimethylamino)-5-hydro xy-2-methyltetrahydro-2H-pyran-3-yl]oxy}-4-hydroxy-2,4-dimethyltetrahydro-2H-pyran-3-yl 3-methylbutanoate
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| DrugBank | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEBI | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.037.140 |
|---|
|
| Formula | C42H69NO15 |
|---|
| Molar mass | 828.006 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
C[C@@H]1C/C=C/C=C/[C@@H]([C@@H](C[C@@H]([C@@H]([C@H]([C@@H](CC(=O)O1)OC(=O)C)OC)O[C@H]2[C@@H]([C@H]([C@@H]([C@H](O2)C)O[C@H]3C[C@@]([C@H]([C@@H](O3)C)OC(=O)CC(C)C)(C)O)N(C)C)O)CC=O)C)O
|
InChI=1S/C42H69NO15/c1-23(2)19-32(47)56-40-27(6)53-34(22-42(40,8)50)57-37-26(5)54-41(36(49)35(37)43(9)10)58-38-29(17-18-44)20-24(3)30(46)16-14-12-13-15-25(4)52-33(48)21-31(39(38)51-11)55-28(7)45/h12-14,16,18,23-27,29-31,34-41,46,49-50H,15,17,19-22H2,1-11H3/b13-12+,16-14+/t24-,25-,26-,27+,29+,30+,31-,34+,35-,36-,37-,38+,39+,40+,41+,42-/m1/s1  Y YKey:XJSFLOJWULLJQS-NGVXBBESSA-N  Y Y
|
 N N Y (what is this?) (verify) Y (what is this?) (verify) |
Josamycin is a macrolide antibiotic. It was isolated by Hamao Umezawa and his colleagues from strains of Streptomyces narbonensis var. josamyceticus var. nova in 1964.[1][2]
It is currently sold in various countries.Brand examples are:
- Europe: Josalid, Josacine, Iosalide, Josamina
- Russia: Wilprafen (Вильпрафен)
- Japan: Josamy
Adverse effects
There has been a case report of edema of the feet.[3]
References
- ^ Osono T, Oka Y, Watanabe S, Okami Y, Umezawa H (July 1967). "A new antibiotic, josamyicn. I. Isolation and physico-chemical characteristics". The Journal of Antibiotics. 20 (3): 174–180. PMID 6072798.
- ^ Umezawa H (1982). "Discovery of josamycin". Giornale Italiano di Chemioterapia. 29 (Suppl 1): 1–10. PMID 6765367.
- ^ Bosch X, Pedrol E, Casado X, Urbano-Marquez A (July 1993). "Josamycin-induced pedal oedema". BMJ. 307 (6895): 26. doi:10.1136/bmj.307.6895.26-a. PMC 1678472. PMID 8343666.
Antibacterials that inhibit protein synthesis (J01A, J01B, J01F, J01G, QJ01XQ) |
|---|
| 30S | Aminoglycosides
(initiation inhibitors) | | -mycin (Streptomyces) | |
|---|
| -micin (Micromonospora) | |
|---|
| other | |
|---|
|
|---|
Tetracycline antibiotics
(tRNA binding) | | Tetracyclines | |
|---|
| Glycylcyclines | |
|---|
|
|---|
|
|---|
| 50S | Oxazolidinone
(initiation inhibitors) |
- Eperezolid
- Linezolid#
- Posizolid
- Radezolid
- Ranbezolid
- Sutezolid
- Tedizolid
|
|---|
| Peptidyl transferase | |
|---|
| MLS (transpeptidation/translocation) | | Macrolides | |
|---|
| Azalides | |
|---|
| Ketolides | |
|---|
| Lincosamides | |
|---|
| Oxepanoprolinamides | |
|---|
| Streptogramins |
- Pristinamycin (IA, IIA, IIB)
- NXL103 (Flopristin, Linopristin)
- Quinupristin/dalfopristin (Dalfopristin, Quinupristin)
- Streptogramin A
- Streptogramin B
- Virginiamycin (S1)
|
|---|
|
|---|
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|

