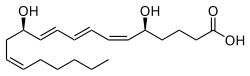
Leukotriene B4
Names
Preferred IUPAC name
(5S ,6Z ,8E ,10E ,12R ,14Z )-5,12-Dihydroxyicosa-6,8,10,14-tetraenoic acid
Identifiers
CAS Number
3D model (JSmol)
ChEBI
ChEMBL
ChemSpider
IUPHAR/BPS
KEGG
UNII
InChI=1S/C20H32O4/c1-2-3-4-5-6-9-13-18(21)14-10-7-8-11-15-19(22)16-12-17-20(23)24/h6-11,14-15,18-19,21-22H,2-5,12-13,16-17H2,1H3,(H,23,24)/b8-7+,9-6-,14-10+,15-11-/t18-,19-/m1/s1
N Key: VNYSSYRCGWBHLG-AMOLWHMGSA-N
N InChI=1/C20H32O4/c1-2-3-4-5-6-9-13-18(21)14-10-7-8-11-15-19(22)16-12-17-20(23)24/h6-11,14-15,18-19,21-22H,2-5,12-13,16-17H2,1H3,(H,23,24)/b8-7+,9-6-,14-10+,15-11-/t18-,19-/m1/s1
Key: VNYSSYRCGWBHLG-AMOLWHMGBE
CCCCC/C=C\C[C@H](/C=C/C=C/C=C\[C@H](CCCC(=O)O)O)O
Properties
Chemical formula
C 20 H 32 O 4
Molar mass
−1
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Leukotriene B4 (LTB4 ) is a leukotriene involved in inflammation. It has been shown to promote insulin resistance in obese mice.
Biochemistry
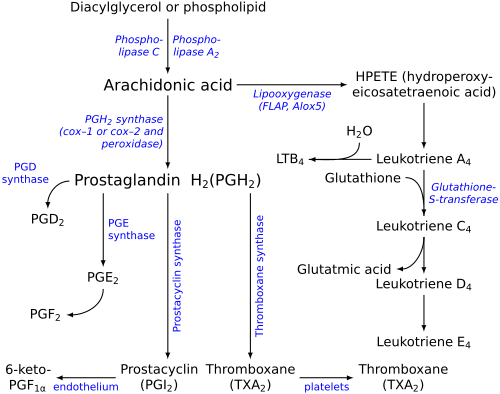
LTB4 is a leukotriene involved in inflammation. It is produced from leukocytes in response to inflammatory mediators and is able to induce the adhesion and activation of leukocytes on the endothelium, allowing them to bind to and cross it into the tissue.[ 1] [ 1] leukotriene A4 .[ 2]
Eicosanoid synthesis (leukotrienes at right)
Diabetes
A study at the University of California, San Diego School of Medicine has shown that LTB4 promotes insulin resistance in obese mice.[ 3] [ 4]
References
^ a b Cotran; Kumar, Collins (1999). Robbins Pathologic Basis of Disease . Philadelphia: W.B Saunders Company. ISBN 0-7216-7335-X . ^ "LTA4H" . uniprot . Retrieved 9 April 2013 .^ "Molecular Link between Obesity and Type 2 Diabetes Reveals Potential Therapy" . UC San Diego Health . Archived from the original on 2022-02-18.^ Li, P; Oh, DY; Bandyopadhyay, G; Lagakos, WS; Talukdar, S; Osborn, O; Johnson, A; Chung, H; Maris, M; Ofrecio, JM; Taguchi, S; Lu, M; Olefsky, JM (2015). "LTB4 promotes insulin resistance in obese mice by acting on macrophages, hepatocytes and myocytes" . Nature Medicine . 21 (3): 239– 247. doi:10.1038/nm.3800 . PMC 4429798 25706874 .
Eicosanoids
Precursor Prostanoids
Prostaglandins (PG)
Thromboxanes (TX)
Leukotrienes (LT)
Precursor
Arachidonic acid 5-hydroperoxide Initial SRS-A
Eoxins (EX)
Precursor
Arachidonic acid 15-hydroperoxide Eoxins
Nonclassic
Lipoxins (LX) (A4 , B4 )
Virodhamine By function
vasoconstriction
vasodilation
platelets: induce
inhibit
leukocytes: induce
inhibit
Leukotriene signaling modulators
Receptor(ligands)
BLT Tooltip Leukotriene B4 receptor
BLT1 Tooltip Leukotriene B4 receptor 1
Antagonists: 20-Carboxy-LTB4 Amelubant
CGS-23131 (LY-223982)
CGS-25019C
CP-105696
CP-195543
Etalocib
LY-293111
Moxilubant
ONO-4057
RG-14893
RP-69698
SB-209247
SC-53228
Ticolubant
U-75302
ZK-158252 BLT2 Tooltip Leukotriene B4 receptor 2
Agonists: 12-HETE12-HHT
12-HpETE
15-HETE
15-HpETE
20-Hydroxy-LTB4
Leukotriene B4 Antagonists: CP-195543LY-255283
ZK-158252
CysLT Tooltip Cysteinyl leukotriene receptor
CysLT1 Tooltip Cysteinyl leukotriene receptor 1
Agonists: Leukotriene C4 Leukotriene D4
Leukotriene E4 Antagonists: AblukastBAYu9773
BAYu9916
BAYx7195
Cinalukast
FPL-55712
ICI-198615
Iralukast
LY-170680
Masilukast
MK-571
Montelukast ONO-1078
Pobilukast
Pranlukast Ritolukast
SKF-104353
SR-2640
Sulukast
Tipelukast
Tomelukast
Verlukast
Zafirlukast ZD-3523
Gemilukast
Quinotolast CysLT2 Tooltip Cysteinyl leukotriene receptor 2
Agonists: Leukotriene C4 Leukotriene D4
Leukotriene E4 Antagonists: BAYu9773BAYu9916 CysLTE Tooltip Cysteinyl leukotriene receptor E
Enzyme(inhibitors)
5-LOX Tooltip Arachidonate 5-lipoxygenase
FLAP Tooltip Arachidonate 5-lipoxygenase-activating protein inhibitors:AM-679
BAYx1005
MK-591
MK-886 12-LOX Tooltip Arachidonate 12-lipoxygenase 15-LOX Tooltip Arachidonate 15-lipoxygenase
2-TEDC
CDC
KNX-100 (SOC-1)
Luteolin PD-146176 LTA4 H Tooltip Leukotriene A4 hydrolase LTB4 H Tooltip Leukotriene B4 ω-hydroxylase LTC4 S Tooltip Leukotriene C4 synthase LTC4 H Tooltip Leukotriene C4 hydrolase LTD4 Tooltip Leukotriene D4 hydrolase
Others
See also
Receptor/signaling modulators Prostanoid signaling modulators
PPARα Tooltip Peroxisome proliferator-activated receptor alpha
Antagonists: GW-6471MK-886 PPARδ Tooltip Peroxisome proliferator-activated receptor delta
Antagonists: FH-535GSK-0660
GSK-3787 PPARγ Tooltip Peroxisome proliferator-activated receptor gamma
SPPARMs Tooltip Selective PPARγ modulator :BADGE EPI-001
INT-131
MK-0533
S26948 Antagonists: FH-535GW-9662
SR-202
T-0070907 Non-selective
Agonists: Ciprofibrate Clinofibrate
Clofibride
Englitazone
Etofibrate
Farglitazar
Netoglitazone
Ronifibrate
Rivoglitazone
Simfibrate
See also
Receptor/signaling modulators