Ciprofibrate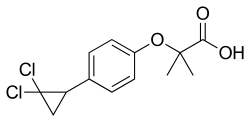 |
|
| AHFS/Drugs.com | International Drug Names |
|---|
| ATC code | |
|---|
|
(RS)-2-[4-(2,2-dichlorocyclopropyl)phenoxy]-2-
methylpropanoic acid
|
| CAS Number | |
|---|
| PubChem CID | |
|---|
| IUPHAR/BPS | |
|---|
| ChemSpider | |
|---|
| UNII | |
|---|
| KEGG | |
|---|
| ChEBI | |
|---|
| ChEMBL | |
|---|
| CompTox Dashboard (EPA) | |
|---|
| ECHA InfoCard | 100.052.478 |
|---|
|
| Formula | C13H14Cl2O3 |
|---|
| Molar mass | 289.15 g·mol−1 |
|---|
| 3D model (JSmol) | |
|---|
| Chirality | Racemic mixture |
|---|
ClC2(Cl)CC2c1ccc(OC(C(=O)O)(C)C)cc1
|
InChI=1S/C13H14Cl2O3/c1-12(2,11(16)17)18-9-5-3-8(4-6-9)10-7-13(10,14)15/h3-6,10H,7H2,1-2H3,(H,16,17)  Y YKey:KPSRODZRAIWAKH-UHFFFAOYSA-N  Y Y
|
| (verify) |
Ciprofibrate is a fibrate that was developed as a lipid-lowering agent.
It was patented in 1972 and approved for medical use in 1985.[1]
References
Lipid-lowering agents (C10) |
|---|
| GI tract | | Cholesterol absorption inhibitors, NPC1L1 | |
|---|
| Bile acid sequestrants/resins (LDL) | |
|---|
|
|---|
| Liver | | Statins (HMG-CoA reductase, LDL) | |
|---|
| Niacin and derivatives (HDL and LDL) | |
|---|
| MTTP inhibitors (VLDL) |
- Dirlotapide
- Lomitapide
- Mitratapide
|
|---|
| ATP citrate lyase inhibitors (LDL) | |
|---|
| Thyromimetics (VLDL) | |
|---|
|
|---|
| Blood vessels | | PPAR agonists (LDL) | |
|---|
| CETP inhibitors (HDL) |
- Anacetrapib†
- Dalcetrapib§
- Evacetrapib§
- Obicetrapib†
- Torcetrapib§
|
|---|
| PCSK9 inhibitors (LDL) |
- Alirocumab
- Bococizumab
- Enlicitide†
- Evolocumab
- Inclisiran
- Lerodalcibep
|
|---|
| ANGPTL3 inhibitors (LDL/HDL) | |
|---|
|
|---|
| Combinations |
- Amlodipine/atorvastatin
- Bempedoic acid/ezetimibe
- Ezetimibe/atorvastatin
- Ezetimibe/rosuvastatin
- Ezetimibe/simvastatin
- Fenofibrate/pravastatin
- Fenofibrate/simvastatin
- Niacin/laropiprant
- Niacin/lovastatin
- Niacin/simvastatin
|
|---|
| Other |
- Alipogene tiparvovec
- Azacosterol
- Azalanstat
- Benfluorex‡
- Darapladib§
- Lapaquistat§
- Magnesium pyridoxal 5-phosphate glutamate
- Meglutol
- Mipomersen
- Olezarsen
- Omega−3-acid ethyl esters
- Plozasiran
- Policosanol
- Probucol
- Tiadenol
- Triparanol‡
- Volanesorsen
|
|---|
- #WHO-EM
- ‡Withdrawn from market
- Clinical trials:
- †Phase III
- §Never to phase III
|
|
|---|
| PPARαTooltip Peroxisome proliferator-activated receptor alpha |
- Antagonists: GW-6471
- MK-886
|
|---|
| PPARδTooltip Peroxisome proliferator-activated receptor delta |
- Antagonists: FH-535
- GSK-0660
- GSK-3787
|
|---|
| PPARγTooltip Peroxisome proliferator-activated receptor gamma |
- SPPARMsTooltip Selective PPARγ modulator: BADGE
- EPI-001
- INT-131
- MK-0533
- S26948
- Antagonists: FH-535
- GW-9662
- SR-202
- T-0070907
|
|---|
| Non-selective |
- Agonists: Ciprofibrate
- Clinofibrate
- Clofibride
- Englitazone
- Etofibrate
- Farglitazar
- Netoglitazone
- Ronifibrate
- Rivoglitazone
- Simfibrate
|
|---|
- See also
- Receptor/signaling modulators
|
