Methallenestril
 | |
| Clinical data | |
|---|---|
| Trade names | Cur-men, Ercostrol, Geklimon, Novestrine, Vallestril (also spelled Vallestrol or Vallestryl) |
| Other names | Methallenoestril; Methallenestrol; Methallenoestrol; Horeau's acid; Allenestrol 6-methyl ether; α,α-Dimethyl-β-ethylallenolic acid 6-methyl ether; β-Ethyl-6-methoxy-α,α-dimethyl-2-naphthalenepropionic acid |
| Routes of administration | By mouth |
| Drug class | Nonsteroidal estrogen |
| ATC code | |
| Identifiers | |
IUPAC name
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.007.485 |
| Chemical and physical data | |
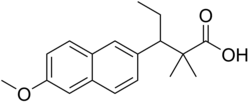
| Formula | C18H22O3 |
| Molar mass | 286.371 g·mol−1 |
| 3D model (JSmol) | |
SMILES
| |
InChI
| |
| (verify) | |
Methallenestril (INN) (brand names Cur-men, Ercostrol, Geklimon, Novestrine, Vallestril), also known as methallenoestril (BAN) and as methallenestrol, as well as Horeau's acid,[1][2] is a synthetic nonsteroidal estrogen and a derivative of allenolic acid and allenestrol (specifically, a methyl ether of it) that was formerly used to treat menstrual issues but is now no longer marketed.[3][4][5][6] It is a seco-analogue of bisdehydrodoisynolic acid, and although methallenestril is potently estrogenic in rats, in humans it is only weakly so in comparison.[7] Vallestril was a brand of methallenestril issued by G. D. Searle & Company in the 1950s.[8] Methallenestril is taken by mouth.[9] By the oral route, a dose of 25 mg methallenestril is approximately equivalent to 1 mg diethylstilbestrol, 4 mg dienestrol, 20 mg hexestrol, 25 mg estrone, 2.5 mg conjugated estrogens, and 0.05 mg ethinylestradiol.[9]
Synthesis
The chemical synthesis has been described:[10] Patent:[11] Unavailable methods:[12][13][14]
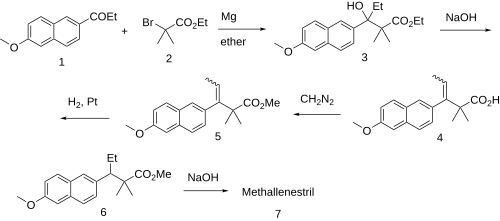
The Grignard reaction between 2-propionyl-6-methoxynaphthalene (promen) [2700-47-2] (1) and Ethyl 2-bromoisobutyrate [600-00-0] (2) occurs to give Ethyl beta-ethyl-beta-hydroxy-6-methoxy-alpha,alpha-dimethylnaphthalene-2-propionate [85536-81-8] (3). Dehydration of the carbinol in aqueous lye may be accompanied by saponification of the ester (although not in the patented version) to give [60533-05-3] (4). Re-esterification with diazomethane gave (5). Catalytic hydrogenation of the olefin led to PC608080 (6). Saponification of the ester completed the synthesis of Methallenestril (7).
An alternative method is described in the patent that relies on 2-cyano-6-methoxynaphthalene (cyanonerolin) [67886-70-8]. The precusor to this is described in a Hoechst patent.[15]
See also
- Carbestrol
- Fenestrel
- Doisynoestrol
- Doisynolic acid
- SC-4289 & allenestrol
References
- ^ Heftmann E (1970). Steroid Biochemistry. Academic Press. p. 144. ISBN 978-0-12-336650-4.
- ^ Dodds EC (March 1949). "Synthetic oestrogens". The Journal of Pharmacy and Pharmacology. 1 (3): 137–147. doi:10.1111/j.2042-7158.1949.tb12391.x. PMID 18114509. S2CID 221921908.
- ^ Ganellin CR, Triggle DJ (21 November 1996). Dictionary of Pharmacological Agents. CRC Press. pp. 1295–. ISBN 978-0-412-46630-4.
- ^ Morton IK, Hall JM (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. pp. 177–. ISBN 978-94-011-4439-1.
- ^ Thomas JA, Keenan EJ (1986). "Estrogens and Estrogenic Compounds". Principles of Endocrine Pharmacology. Springer Science & Business Media. p. 136. ISBN 978-1-4684-5036-1.
- ^ Herbai G, Ljunghall S (1983). "Normalization of hypercalcaemia of primary hyperparathyroidism by treatment with methallenestril, a synthetic oestrogen with low oestrogenicity". Urologia Internationalis. 38 (6): 371–373. doi:10.1159/000280925. PMID 6659184.
- ^ Kirk RE, Othmer DF (1980). Encyclopedia of chemical technology. Wiley. p. 670. ISBN 978-0-471-02065-3.
- ^ Catalog of Copyright Entries: Third Series. Vol. 17. Copyright Office, Library of Congress. July–December 1963. pp. 1984–.
- ^ a b Swyer GI (April 1959). "The oestrogens". British Medical Journal. 1 (5128): 1029–1031. doi:10.1136/bmj.1.5128.1029. PMC 1993181. PMID 13638626.
- ^ Wieland, P.; Miescher, K. (January 1948). "Abkömmlinge alkylierter β‐Naphtyl‐valeriansäuren. Über oestrogene Carbonsäuren XXVI". Helvetica Chimica Acta. 31 (6): 1844–1854. doi:10.1002/hlca.19480310650.
- ^ Horeau Alain & Jacques Jean, U.S. patent 2,547,123 (1951 to Individual).
- ^ Gay, R. et al, Bull. Soc. Chim. Fr., 1955, 955.
- ^ Jacques; Horeau Bulletin de la Societe Chimique de France, 1948 , p. 711,715.
- ^ Dubois,J.C. et al. Bulletin de la Societe Chimique de France, 1967 , p. 1827 – 1833.
- ^ Theodor Papenfuhs & Kurt Gengnagel, U.S. patent 4,137,272 (1979 to Hoechst AG).