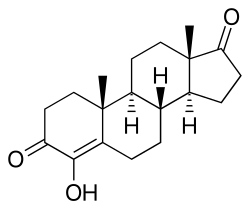
Formestane Trade names Lentaron, others Other names 4-Hydroxyandrost-4-ene-3,17-dione AHFS/Drugs.com International Drug Names Routes of Intramuscular injection Drug class Aromatase inhibitor; Antiestrogen ATC code
(8R ,9S ,10R ,13S ,14S )-4-hydroxy-10,13-dimethyl-2,6,7,8,9,11,12,14,15,16-decahydro-1H -cyclopenta[a ]phenanthrene-3,17-dione
CAS Number PubChem CID ChemSpider UNII KEGG ChEBI ChEMBL CompTox Dashboard (EPA ) ECHA InfoCard 100.153.838 Formula C 19 H 26 O 3 Molar mass −1 3D model (JSmol)
O=C4C(/O)=C3/CC[C@@H]2[C@H](CC[C@@]1(C(=O)CC[C@H]12)C)[C@@]3(C)CC4
InChI=1S/C19H26O3/c1-18-10-8-15(20)17(22)14(18)4-3-11-12-5-6-16(21)19(12,2)9-7-13(11)18/h11-13,22H,3-10H2,1-2H3/t11-,12-,13-,18+,19-/m0/s1
Y Key:OSVMTWJCGUFAOD-KZQROQTASA-N
Y N Y (verify)
Formestane , formerly sold under the brand name Lentaron among others, is a steroidal, selective aromatase inhibitor which is used in the treatment of estrogen receptor-positive breast cancer in postmenopausal women.[ 1] [ 2] androstenedione .
Formestane is often used to suppress the production of estrogens from anabolic steroids or prohormones. It also acts as a prohormone to 4-hydroxytestosterone, an active steroid which displays weak androgenic activity in addition to acting as a weak aromatase inhibitor.
Pharmacodynamics of aromatase inhibitors
Generation
Medication
Dosage
% inhibitiona
Classb
IC50 c
First
Testolactone 250 mg 4x/day p.o.
?
Type I
?
100 mg 3x/week i.m.
?
Rogletimide
200 mg 2x/day p.o. p.o. p.o.
50.6%
Type II
?
Aminoglutethimide 250 mg mg 4x/day p.o.
90.6%
Type II
4,500 nM
Second
Formestane
125 mg 1x/day p.o. p.o. p.o.
72.3%
Type I
30 nM
250 mg 1x/2 weeks i.m. i.m. i.m.
84.8%
Fadrozole
1 mg 1x/day p.o. p.o.
82.4%
Type II
?
Third
Exemestane 25 mg 1x/day p.o.
97.9%
Type I
15 nM
Anastrozole 1 mg 1x/day p.o. p.o.
96.7–97.3%
Type II
10 nM
Letrozole 0.5 mg 1x/day p.o. p.o.
98.4%
Type II
2.5 nM
Footnotes: a = In postmenopausal women. b = Type I: Steroidal, irreversible (substrate-binding site). Type II: Nonsteroidal, reversible (binding to and interference with the cytochrome P450 heme moiety). c = In breast cancer homogenates. Sources: See template.
Chemistry
Synthesis
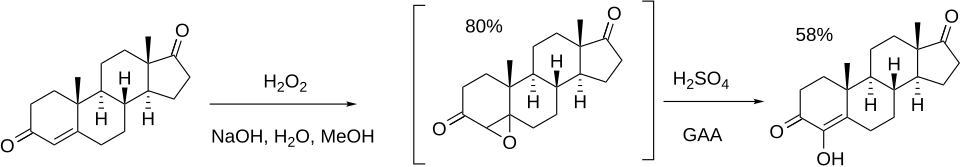
The synthesis of formestane was reported:[ 3] [ 4] [ 5]
500pxclass=skin-invert-image Reaction of androstenedione with alkaline hydrogen peroxide gives the 4,5-epoxyandrosta-3,17-dione, PC13633174. Addition of acid completes the synthesis of formestane.
References
^ Pérez Carrión R, Alberola Candel V, Calabresi F, et al. (1994). "Comparison of the selective aromatase inhibitor formestane with tamoxifen as first-line hormonal therapy in postmenopausal women with advanced breast cancer". Ann. Oncol . 5 (Suppl 7): S19–24. PMID 7873457 . ^ "Formestane" .^ Angela M. H. Brodie, Harry J. Brodie, & David A. Marsh, U.S. patent 4,235,893
^ Jr Roy H Bible & Placek Chester, US2908682 (1959 to GD Searle LLC).
^ Franco Faustini, US4757061 (1988 to Pfizer Italia SRL).
Estrogens and antiestrogens
Estrogens
ER Tooltip Estrogen receptor agonists
Steroidal: AlfatradiolCertain androgens/anabolic steroids (e.g., testosterone , testosterone esters, methyltestosterone , metandienone , nandrolone esters) (via estrogenic metabolites)
Certain progestins (e.g., norethisterone , noretynodrel , etynodiol diacetate , tibolone)
Clomestrone
Cloxestradiol acetate
Conjugated estriol
Conjugated estrogens Epiestriol Epimestrol
Esterified estrogens
Estetrol†
Estradiol
Estradiol esters (e.g., estradiol acetate, estradiol benzoate, estradiol cypionate, estradiol enanthate, estradiol undecylate, estradiol valerate, polyestradiol phosphate, estradiol ester mixtures (Climacteron))
Estramustine phosphate
Estriol Estriol esters (e.g., estriol succinate, polyestriol phosphate)
Estrogenic substances
Estrone Estrone esters
Estrone sulfate
Estropipate (piperazine estrone sulfate)
Ethinylestradiol #
Ethinylestradiol sulfonate Hydroxyestrone diacetate
Mestranol Methylestradiol
Moxestrol
Nilestriol
Prasterone (dehydroepiandrosterone; DHEA)
Promestriene
Quinestradol
Quinestrol Progonadotropins
Antiandrogens (e.g., bicalutamide )
GnRH agonists (e.g., GnRH (gonadorelin), leuprorelin )Gonadotropins (e.g., FSH Tooltip follicle-stimulating hormone , LH Tooltip luteinizing hormone )
Antiestrogens
ER Tooltip Estrogen receptor antagonistsSERMs Tooltip selective estrogen receptor modulators /SERDs Tooltip selective estrogen receptor downregulators )
Exclusively antagonistic: ElacestrantFulvestrant Imlunestrant Aromatase inhibitors Antigonadotropins
Androgens/anabolic steroids (e.g., testosterone , testosterone esters, nandrolone esters, oxandrolone , fluoxymesterone )
D2 receptor antagonists (prolactin releasers) (e.g., domperidone , metoclopramide , risperidone , haloperidol , chlorpromazine , sulpiride )
GnRH agonists (e.g., leuprorelin , goserelin )GnRH antagonists (e.g., cetrorelix , elagolix)Progestogens (e.g., chlormadinone acetate, cyproterone acetate, gestonorone caproate, hydroxyprogesterone caproate, medroxyprogesterone acetate , megestrol acetate) Others
# WHO-EM‡ Withdrawn from marketClinical trials:
† Phase III§ Never to phase III
See also
Estrogen receptor modulators
Androgens and antiandrogens
Progestogens and antiprogestogens
List of estrogens
Androgen receptor modulators
AR Tooltip Androgen receptor
Agonists SARMs Tooltip Selective androgen receptor modulator
Nonsteroidal: 198RL26ACP-105
AC-262,536
Acetothiolutamide
Acetoxolutamide
Andarine (acetamidoxolutamide, androxolutamide, GTx-007, S-4)
BMS-564,929
DTIB
Enobosarm (ostarine, MK-2866, GTx-024, S-22)
FTBU-1
GLPG-0492
GSK2881078
GSK-4336A
GSK-8698
LG121071 (LGD-121071)
LGD-2226
LGD-2941 (LGD-122941)
LGD-3303
LGD-4033
LY305
JNJ-26146900
JNJ-28330835
JNJ-37654032
OPK-88004 (LY-2452473, TT-701)
ORM-11984
PF-06260414
R-1
RU-59063
S-1
S-23
S-40503
S-101479
Vosilasarm Steroidal: EM-9017MK-0773
S42
TFM-4AS-1
YK-11 Antagonists
Steroidal: 7α-Thioprogesterone7α-Thiospironolactone
7α-Thiomethylspironolactone
11α-Hydroxyprogesterone
15β-Hydroxycyproterone acetate
Abiraterone
Abiraterone acetate
Allyltestosterone
Benorterone
BOMT
Canrenoic acid
Canrenone
Chlormadinone acetate
Clascoterone
Clometerone
Cyproheptadine Cyproterone Cyproterone acetate
Delanterone
Delmadinone acetate
Dicirenone
Dienogest
Drospirenone DU-41165
Edogestrone
EM-4350
EM-5854
EM-5855
EM-6537
Epitestosterone
Galeterone
Guggulsterone
Ludaterone
Medrogestone
Megestrol acetate
Mespirenone
Metogest
Mexrenone
Mifepristone Nomegestrol acetate
Nordinone
Osaterone
Osaterone acetate
Oxendolone
Potassium canrenoate
Promegestone Prorenone
Rosterolone
RU-15328
SC-5233 (spirolactone)
Spironolactone Spirorenone
Spiroxasone
Topterone
Trimegestone
Trimethyltrienolone (R-2956)
Zanoterone
GPRC6A
See also
Receptor/signaling modulators
Androgens and antiandrogens
Estrogen receptor modulators
Progesterone receptor modulators
List of androgens and anabolic steroids