Nickel(II) thiocyanate
 Sample of nickel(II) thiocyanate
| |
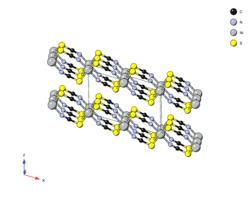 Crystal structure of nickel(II) thiocyanate
| |
| Identifiers | |
|---|---|
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.033.808 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Ni(SCN)2 |
| Molar mass | 174.86 g/mol[1] |
| Appearance | green-brown powder |
| Density | 2.59 g/cm3[1] |
| Melting point | decomposes[1] |
Magnetic susceptibility (χ)
|
5×10−3 cm3/mol[2] |
| Structure | |
Crystal structure
|
Hg(SCN)2 structure |
Coordination geometry
|
Octahedral |
| Hazards | |
| GHS labelling: | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H317, H334, H341, H350i, H360D, H372, H410 |
Precautionary statements
|
P201, P202, P260, P264, P270, P272, P273, P280, P281, P285, P302+P352, P304+P341, P308+P313, P314, P321, P333+P313, P342+P311, P363, P391, P405, P501 |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Nickel(II) thiocyanate is a coordination polymer with formula Ni(SCN)2. It is a green-brown solid and its crystal structure was determined first in 1982.[1]
Structure
The structure of Ni(SCN)2 was determined via single-crystal X-ray diffraction and consists of two-dimensional sheets held together through Van der Waals forces. It belongs to mercury thiocyanate structure-type and can be considered a distorted form of the NiBr2 (CdI2) structure. Each nickel is octahedrally coordinated by four sulfurs and two nitrogens. The sulfur end of the SCN− ligand is doubly bridging.[1]
Synthesis
Nickel(II) thiocyanate can be prepared via the reaction of barium thiocyanate and nickel sulfate solutions. After removal of the precipitated barium sulfate, the solution is allowed to evaporate leaving microcrystalline Ni(SCN)2.[3][4]
Magnetism
Nickel(II) thiocyanate, like nickel(II) iodide, nickel(II) bromide and nickel(II) chloride, is an antiferromagnet at low temperatures.[2]
References
- ^ a b c d e Dubler, Erich; Relier, Armin; Oswald, H. R. (1982-01-01). "Intermediates in thermal decomposition of nickel(II) complexes: The crystal structures of Ni(SCN)2(NH3)2 and Ni(SCN)2". Zeitschrift für Kristallographie – Crystalline Materials. 161 (1–4): 265–278. doi:10.1524/zkri.1982.161.14.265. ISSN 2196-7105. S2CID 201671776.
- ^ a b DeFotis, G. C.; Dell, K. D.; Krovich, D. J.; Brubaker, W. W. (1993-05-15). "Antiferromagnetism of Ni(SCN)2". Journal of Applied Physics. 73 (10): 5386–5388. Bibcode:1993JAP....73.5386D. doi:10.1063/1.353740. ISSN 0021-8979.
- ^ Geers, Madeleine; Jarvis, David M.; Liu, Cheng; Saxena, Siddharth S.; Pitcairn, Jem; Myatt, Emily; Hallweger, Sebastian A.; Kronawitter, Silva M.; Kieslich, Gregor; Ling, Sanliang; Cairns, Andrew B.; Daisenberger, Dominik; Fabelo, Oscar; Cañadillas-Delgado, Laura; Cliffe, Matthew J. (2023). "High-pressure behavior of the magnetic van der Waals molecular framework Ni(NCS)2". Physical Review B. 108 (14) 144439. arXiv:2309.04477. Bibcode:2023PhRvB.108n4439G. doi:10.1103/PhysRevB.108.144439.
- ^ Bassey, Euan N.; Paddison, Joseph A. M.; Keyzer, Evan N.; Lee, Jeongjae; Manuel, Pascal; Da Silva, Ivan; Dutton, Siân E.; Grey, Clare P.; Cliffe, Matthew J. (2020). "Strengthening the Magnetic Interactions in Pseudobinary First-Row Transition Metal Thiocyanates, M(NCS)2". Inorganic Chemistry. 59 (16): 11627–11639. doi:10.1021/acs.inorgchem.0c01478. PMID 32799496.