Nickel(II) titanate
 | |
| Names | |
|---|---|
| IUPAC name
Nickel(IV) titanate
| |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.031.647 |
| EC Number |
|
PubChem CID
|
|
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
NiTiO3 |
| Molar mass | 154.61 g/mol |
| Appearance | yellow powder |
| Density | 4.44 g/cm3 |
Solubility in water
|
0.0396 g/100 mL |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
 
|
Signal word
|
Danger |
Hazard statements
|
H317, H350i, H372 |
Precautionary statements
|
P203, P260, P264, P270, P272, P280, P302+P352, P318, P319, P321, P333+P317, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 
3
0
0 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Nickel(II) titanate is an inorganic compound with the chemical formula NiTiO3. It is a coordination compound between nickel(II), titanium(IV) and oxide ions. It has the appearance of a yellow powder.
Structure
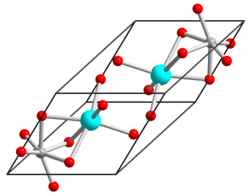
Nickel(II) titanate crystallizes at 600 °C[2] and is stable at room temperature and normal pressure in an ilmenite structure with rhombohedral R3 symmetry.[3] It consists of alternating layers of Ni and Ti along the rhombohedral axis with O layers between them.[2]
Another description of nickel(II) titanite's illemite structure consists of a pseudo close packed hexagonal array of O2− ions with two thirds occupied by an ordered hexagonal like cation.[4] The average crystallite's size was estimated at 42 nm with lattice constants of a = 5.032 Å, b = 5.032 Å, c = 4.753 Å.[4]
Preparation
Nickel(II) titanate can be prepared by the autocombustion of a mixture of titanium isopropoxide, nickel(II) nitrate (oxidizer), and alanine (fuel) in isopropyl alcohol solution.[2] The resulting dark green product is then calcined.[2]
Another method involves heating NiO and TiO2 at 1350 °C for three hours.[5]
- NiO + TiO2 → NiTiO3
NiTiO3 residue is formed in the thermal decomposition of the heterobimetallic complex Ni2Ti2(OEt)2(μ-OEt)6(2,4-pentanedionate)4.[4]
Applications
Nickel(II) titanate is used as a yellow pigment.[6] It has been used as a catalyst for toluene oxidation.[2]
It is of research interest as an electronic material due to its high dielectric constant (40), weak magnetism, and semiconductivity.[7] It has received attention as a possible candidate for multiferroic materials capable of magnetization through the application of an electric field.[8]
References
- ^ PubChem. "Nickel titanium oxide". pubchem.ncbi.nlm.nih.gov. Retrieved 2026-01-25.
- ^ a b c d e Traistaru, G.A. (July 2011). "Synthesis and Characterization of NiTiO3 and NiFe2O4 as catalysts for Toluene Oxidation" (PDF). Digest Journal of Nanomaterials and Biostructures.
- ^ Shirane, Gen; Pickart, S. J.; Ishikawa, Yoshikazu (1959-10-15). "Neutron Diffraction Study of Antiferromagnetic MnTiO3 and NiTiO3". Journal of the Physical Society of Japan. 14 (10): 1352–1360. Bibcode:1959JPSJ...14.1352S. doi:10.1143/JPSJ.14.1352. ISSN 0031-9015.
- ^ a b c Tahir, Asif Ali; Mazhar, Muhammad; Hamid, Mazhar; Wijayantha, K.G. Upul; Molloy, Kieran C. (2009-05-06). "Photooxidation of water by NiTiO3 deposited from single source precursor [Ni2Ti2(OEt)2(μ-OEt)6(acac)4] by AACVD". Dalton Transactions (19): 3674–3680. doi:10.1039/B818765G. ISSN 1477-9234. PMID 19417932.
- ^ Lerch, M.; Laqua, W. (1992-04-01). "Beiträge zu den Eigenschaften von Titanaten mit Ilmenitstruktur. II. Zur Thermodynamik und elektrischen Leitfähigkeit von NiTiO3 und anderen oxidischen Phasen mit Ilmenitstruktur". Zeitschrift für Anorganische und Allgemeine Chemie. 610 (4): 57–63. doi:10.1002/zaac.19926100110. ISSN 1521-3749.
- ^ Jose, Sheethu; Joshy, Deepak; Narendranath, Soumya B.; Periyat, Pradeepan (2019-06-01). "Recent advances in infrared reflective inorganic pigments". Solar Energy Materials and Solar Cells. 194: 21. doi:10.1016/j.solmat.2019.01.037. ISSN 0927-0248.
- ^ Zhang, Xiaochao; Lu, Bingqian; Li, Rui; Fan, Caimei; Liang, Zhenhai; Han, Peide (2015-11-01). "Structural, electronic and optical properties of Ilmenite ATiO3(A=Fe, Co, Ni)". Materials Science in Semiconductor Processing. 39: 6–16. doi:10.1016/j.mssp.2015.04.041. ISSN 1369-8001.
- ^ Varga, Tamas; Droubay, Timothy C.; Bowden, Mark E.; Nachimuthu, Ponnusamy; Shutthanandan, Vaithiyalingam; Bolin, Trudy B.; Shelton, William A.; Chambers, Scott A. (2012-06-30). "Epitaxial growth of NiTiO3 with a distorted ilmenite structure". Thin Solid Films. 520 (17): 5534–5541. Bibcode:2012TSF...520.5534V. doi:10.1016/j.tsf.2012.04.060.