Sodium propionate
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Sodium propanoate | |
| Other names
Sodium propionate
Napropion E281 | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.004.810 |
| EC Number |
|
| E number | E281 (preservatives) |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C3H5NaO2 |
| Molar mass | 96.060 g/mol |
| Appearance | Transparent crystals |
| Odor | faint acetic-butyric odor |
| Melting point | 289 °C (552 °F; 562 K) |
Solubility in water
|
1 g/mL |
| Solubility in ethanol | 41.7 g/L |
| Pharmacology | |
ATC code
|
S01AX10 (WHO) QA16QA02 (WHO) |
| Hazards | |
| GHS labelling:[2] | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H302, H317, H318, H411 |
Precautionary statements
|
P261, P264, P264+P265, P270, P272, P273, P280, P301+P317, P302+P352, P305+P354+P338, P317, P321, P330, P333+P317, P362+P364, P391, P501 |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
6332 mg/kg (mouse, oral)[2] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Sodium propanoate or sodium propionate is the sodium salt of propionic acid which has the chemical formula Na(C2H5COO). This white crystalline solid is deliquescent in moist air.[2]
Structure
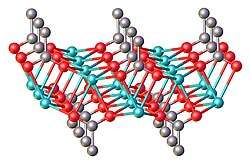
Anhydrous sodium propionate is a polymeric structure, featuring trigonal prismatic Na+ centers bonded to six oxygen ligands provided by the carboxylates. A layered structure is observed, with the hydrophobic ethyl groups projecting into the layered galleries. With hydrated sodium propionate, some of these Na-carboxylate linkages are displaced by water.
Preparation
It is produced by the reaction of propionic acid and sodium carbonate or sodium hydroxide.[4]
Uses
It is used as a food preservative and is represented by the food labeling E number E281 in Europe; it is used primarily as a mold inhibitor in bakery products. It is approved for use as a food additive in the EU,[5] USA[6] and Australia and New Zealand[7] (where it is listed by its INS number 281).
Reactions
Decomposition takes place via ketonization, yielding the symmetric ketone (3-pentanone) and sodium carbonate:[4]
- 2Na(O2CEt) → Na2CO3 + Et(CO)Et
Some side reactions resulting in the release of carbon dioxide are observed.[4]
See also
- Propionic acid, E 280
- Calcium propionate, E 282
- Potassium propionate, E 283
References
- ^ Merck Index, 11th Edition, 8623.
- ^ a b c PubChem. "Sodium Propionate". pubchem.ncbi.nlm.nih.gov. Retrieved 2026-02-01.
- ^ Fábry, Jan; Samolová, Erika (2020). "Layered alkali propanoates M+(C2H5COO)−; M+= Na+, K+, Rb+, Cs+". Acta Crystallographica Section E. 76 (9): 1508–1513. Bibcode:2020AcCrE..76.1508F. doi:10.1107/S2056989020011469. PMC 7472758. PMID 32939309.
- ^ a b c Grivel, J. -C. (2018-11-01). "New insights into the thermal behavior and decomposition of sodium propionate". Journal of Analytical and Applied Pyrolysis. 136: 62–68. doi:10.1016/j.jaap.2018.10.023. ISSN 0165-2370.
- ^ UK Food Standards Agency: "Current EU approved additives and their E Numbers". Retrieved 2011-10-27.
- ^ US Food and Drug Administration: "Listing of Food Additives Status Part II". Food and Drug Administration. Archived from the original on January 8, 2010. Retrieved 2011-10-27.
- ^ Australia New Zealand Food Standards Code"Standard 1.2.4 - Labelling of ingredients". 8 September 2011. Retrieved 2011-10-27.
External links
- Sodium propanoate at Sci-toys.com