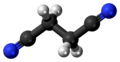
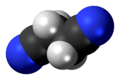
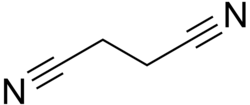
Succinonitrile
 | |||
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Butanedinitrile[1] | |||
Other names
| |||
| Identifiers | |||
CAS Number
|
|||
3D model (JSmol)
|
|||
Beilstein Reference
|
1098380 | ||
| ChemSpider | |||
| ECHA InfoCard | 100.003.441 | ||
| EC Number |
| ||
| MeSH | succinonitrile | ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
C4H4N2 | ||
| Molar mass | 80.090 g·mol−1 | ||
| Appearance | Colorless, waxy crystals | ||
| Odor | odorless[2] | ||
| Density | 985 mg mL−1 | ||
| Melting point | 58 °C (136 °F; 331 K)[3] | ||
| Boiling point | 266.1 °C; 510.9 °F; 539.2 K | ||
Solubility in water
|
130 g L−1 | ||
| Vapor pressure | 300 Pa (at 100 °C) | ||
| Thermochemistry | |||
Heat capacity (C)
|
145.60 J K−1 mol−1 | ||
Std molar
entropy (S⦵298) |
191.59 J K−1 mol−1 | ||
Std enthalpy of
formation (ΔfH⦵298) |
139.3–140.4 kJ mol−1 | ||
Std enthalpy of
combustion (ΔcH⦵298) |
−2.2848–−2.2860 MJ mol−1 | ||
| Hazards | |||
| GHS labelling: | |||
Pictograms
|

| ||
Signal word
|
Warning | ||
Hazard statements
|
H302, H315, H319, H335 | ||
Precautionary statements
|
P261, P305+P351+P338 | ||
| Flash point | 113 °C (235 °F; 386 K) | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
450 mg kg−1 (oral, rat) | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
none[2] | ||
REL (Recommended)
|
TWA 6 ppm (20 mg/m3)[2] | ||
IDLH (Immediate danger)
|
N.D.[2] | ||
| Related compounds | |||
Related alkanenitriles
|
|||
Related compounds
|
DBNPA | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
Succinonitrile, also butanedinitrile, is a nitrile, with the formula of C2H4(CN)2. It is an odorless, colorless to light brown crystal or colourless waxy solid which melts at 58 °C.[4][5]
Succinonitrile is produced by the addition of hydrogen cyanide to acrylonitrile (hydrocyanation):[6]
- CH2=CHCN + HCN → NCCH2CH2CN
Hydrogenation of succinonitrile yields putrescine (1,4-diaminobutane).
Applications
Succinonitrile may find applications as the "solvent" (while still in its waxy state) in lithium batteries.
Safety
Like most nitriles, succinonitrile is toxic and can release cyanide ions in the body. NIOSH recommends a TWA exposure limit of 6 ppm (20 mg/m³).[4]
See also
- Malononitrile - A di-nitrile with 3 carbon atoms
- Glutaronitrile - A di-nitrile with 5 carbon atoms
- Adiponitrile - A di-nitrile with 6 carbon atoms
References
- ^ International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 902. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ a b c d NIOSH Pocket Guide to Chemical Hazards. "#0573". National Institute for Occupational Safety and Health (NIOSH).
- ^ Rubinstein, E. R.; Tirmizi, S. H.; Glicksman, M. E. (1990-11-01). "Long-term purity assessment in succinonitrile". Journal of Crystal Growth. 106 (1): 89–96. Bibcode:1990JCrGr.106...89R. doi:10.1016/0022-0248(90)90290-2. ISSN 0022-0248.
- ^ a b "SUCCINONITRILE | CAMEO Chemicals | NOAA". cameochemicals.noaa.gov. Retrieved 2025-11-21.
- ^ PubChem. "Hazardous Substances Data Bank (HSDB) : 7190". pubchem.ncbi.nlm.nih.gov. Retrieved 2025-12-22.
- ^ "Nitriles". Ullmann's Encyclopedia of Industrial Chemistry (7th ed.). Retrieved 2007-09-10.
External links