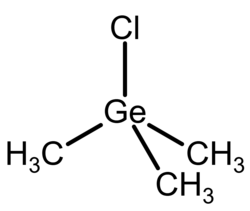
Trimethylgermanium chloride
 | |
| Names | |
|---|---|
| IUPAC name
Chloro(trimethyl)germane
| |
Other names
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
(CH3)3GeCl |
| Molar mass | 153.19 g·mol−1 |
| Appearance | Colorless liquid[1] |
| Density | 1.24 g/cm3[1] |
| Melting point | −13 °C (9 °F; 260 K)[1] |
| Boiling point | 102 °C (216 °F; 375 K)[1] |
Solubility in water
|
Insoluble[2] |
| Solubility | Miscible with diethyl ether, tetrahydrofuran, hexanes, benzene, dichloromethane and chloroform.[2] |
Refractive index (nD)
|
1.433[2] |
| Structure | |
Molecular shape
|
Tetrahedral at Ge |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Serious eye damage |
| GHS labelling: | |
Pictograms
|
 
|
Signal word
|
Danger |
Hazard statements
|
H225, H314 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P260, P264, P280, P301+P330+P331, P302+P361+P354, P303+P361+P353, P304+P340, P305+P354+P338, P316, P321, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 
3
3
1 |
| Flash point | 1 °C (34 °F)[1] |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Trimethylgermanium chloride is an organogermanium compound with the chemical formula (CH3)3GeCl. It is a colorless liquid.
Synthesis
Trimethylgermanium chloride can be synthesized in high yield by reacting tetramethylgermanium with hydrogen chloride in the presence of aluminium trichloride as a catalyst.[3]
- Ge(CH3)4 + HCl → (CH3)3GeCl + CH4
It can also be synthesized by reaction of tetramethylgermanium with dimethylgermanium dichloride in a 1:1 molar ratio, with methylgermanium trichloride in a 2:1 molar ratio, or with germanium tetrachloride in a 3:1 molar ratio, in the presence of gallium trichloride as a catalyst.[4]
- Ge(CH3)4 + (CH3)2GeCl2 → 2 (CH3)3GeCl
- 2 Ge(CH3)4 + CH3GeCl3 → 3 (CH3)3GeCl
- 3 Ge(CH3)4 + GeCl4 → 4 (CH3)3GeCl
Uses
Trimethylgermanium chloride can be used to make trimethyl(2-thienyl)germane. It is also used as a methylating agent and precursor to prepare Grignard reagents in organic synthesis. It can be used as a precursor to germanium-masked dienolates for regioselective C-C bond formation, germanium enolates and α-germanate esters for aldol condensation reactions, Claisen rearrangements, and Peterson-type reactions.[2]
Reactions
Trimethylgermanium chloride is sensitive to moisture. It reacts slowly with water, producing trimethylgermanol and hydrogen chloride.[2]
- (CH3)3GeCl + H2O → (CH3)3GeOH + HCl
It reacts with tris(trimethylsilyl)antimony to give tris(trimethylgermyl)antimony and trimethylsilyl chloride.[5]
- 3 (CH3)3GeCl + ((CH3)3Si)3Sb → ((CH3)3Ge)3Sb + 3 (CH3)3SiCl
Safety
When it burns, it releases toxic and irritating smoke and gases including carbon monoxide, carbon dioxide, hydrogen chloride, germanium monoxide and germanium dioxide.[1]
References
- ^ a b c d e f "Chlorotrimethylgermane". www.sigmaaldrich.com. Retrieved February 27, 2026.
- ^ a b c d e "TRIMETHYLGERMANIUM CHLORIDE | 1529-47-1". ChemicalBook. Retrieved 2026-02-27.
- ^ Houben-Weyl Methods of Organic Chemistry Vol. XIII/6, 4th Edition: Organogermanium- and -tin Compounds. Georg Thieme Verlag. 14 May 2014. ISBN 978-3-13-180734-2.
- ^ Schumann-Ruidisch, I.; Lieb, V.; Jutzi-Mebert, B. (1967). "Galliumchlorid-katalysierte Konproportionierungsreaktionen von Alkylgermaniumhalogeniden". Zeitschrift für Anorganische und Allgemeine Chemie. 355 (1–2): 64–72. doi:10.1002/zaac.19673550108.
- ^ Ates, M.; Breunig, H. J.; Denker, M. (1995). "FORMATION OF (Me3M)3Sb (M = Ge, Sn, Pb) AND (Me3M)4Sb2(M = Pb) BY REACTION OF (Me3Si)3Sb WITH Me3MCL". Phosphorus, Sulfur, and Silicon and the Related Elements. 102 (1–4): 287–289. doi:10.1080/10426509508042569.