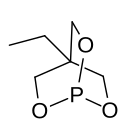
Trimethylolpropane phosphite
|
|
| Names
|
Preferred IUPAC name
4-Ethyl-2,6,7-trioxa-1-phosphabicyclo[2.2.2]octane |
| Other names
EtCage; Ethyl bicyclic phosphite; Trishydroxymethylpropane bicyclic phosphite
|
| Identifiers
|
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider
|
|
| ECHA InfoCard
|
100.011.385
|
| EC Number
|
|
|
|
|
| UNII
|
|
|
|
|
InChI=1S/C6H11O3P/c1-2-6-3-7-10(8-4-6)9-5-6/h2-5H2,1H3  N NKey: QRUSNTDXJQBKBI-UHFFFAOYSA-N  N NInChI=1/C6H11O3P/c1-2-6-3-7-10(8-4-6)9-5-6/h2-5H2,1H3 Key: QRUSNTDXJQBKBI-UHFFFAOYAD
|
|
|
| Properties
|
Chemical formula
|
C6H11O3P
|
| Molar mass
|
162.125 g·mol−1
|
| Appearance
|
white waxy solid
|
| Melting point
|
56 °C (133 °F; 329 K)
|
Solubility in water
|
organic solvents
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
|
Trimethylolpropane phosphite, C2H5C(CH2O)3P, is a phosphite ester used as a ligand in organometallic chemistry. Trimethylolpropane phosphite is sometimes abbreviated to EtCage. It is a white solid that is soluble in organic solvents. It is also highly toxic.[1]
Preparation and reactions
It is prepared by reaction of trimethylolpropane with phosphorus trichloride or by transesterification with trimethylphosphite:[2]
- P(OMe)3 + EtC(CH2OH)3 → 3 MeOH + EtC(CH2O)3P
The first member of this series was derived from trimethylolethane,[3] but these derivatives are often poorly soluble. For this reason, the ethyl derivative has received more attention.[4]
Reactions
The compound forms an isolable ozonide, which degrades above 0 °C to release singlet O2.[1]
Coordination chemistry
Several EtCage complexes are known, since the ligand is highly basic (for a phosphite) and has a small ligand cone angle (101°). Illustrative complexes include [(EtCage)2Mo(CO)4], [Ir4(CO)11(EtCage)] and (CpMe5)RuCl(EtCage)2, shown below.
2Mo(CO)4-from-xtal-2007-3D-balls.png)
11(EtCage)-from-xtal-1993-3D-balls.png)
RuCl(EtCage)2-from-xtal-1996-3D-balls.png)
Safety
Trimethylolpropane phosphite is very toxic and is a convulsant. LD50 is 1.1 mg per kg bodyweight (mice, i.p.).[5][6]
References
- ^ a b Ende, Christopher; Parker, Kathlyn (2014). "4-Ethyl-2,6,7-trioxa-1-phosphabicyclo[2.2.2]octane". e-EROS Encyclopedia of Reagents for Organic Synthesis. pp. 1–3. doi:10.1002/047084289X.rn01781. ISBN 9780470842898.
- ^ Verkade, J. G. "Spectroscopic studies of metal-phosphorus bonding in coordination complexes" Coordination Chemistry Reviews 1972, vol. 9, 1-106. doi:10.1016/S0010-8545(00)80224-6
- ^ Verkade, J. G.; Reynolds, L. T. "The synthesis of a novel ester of phosphorus and of arsenic" Journal of Organic Chemistry (1960), 25, 663-5. doi:10.1021/jo01074a622
- ^ Huttemann, T. J., Jr.; Foxman, B. M.; Sperati, C. R.; Verkade, J. G. "Transition metal complexes of a constrained phosphite ester. IV. Compounds of cobalt(I), cobalt(III), nickel(II), and nickel(0)" Inorganic Chemistry (1965), 4(7), 950-3. doi:10.1021/ic50029a005
- ^ Ralf Stöhr et al. Chemische Kampfstoffe und Schutz vor chemischen Kampfstoffen 2. Aufl. Militärverlag der DDR, 1985 (german)
- ^ Milbrath, Dean S.; Engel, Judith L.; Verkade, John G.; Casida, John E. (1979). "Structure-toxicity relationships of 1-substituted-4-alkyl-2,6,7-trioxabicyclo[2.2.2.]octanes". Toxicology and Applied Pharmacology. 47 (2): 287–93. Bibcode:1979ToxAP..47..287M. doi:10.1016/0041-008X(79)90323-5. PMID 452023.
Convulsants |
|---|
| GABA receptor antagonists | |
|---|
| GABA synthesis inhibitors | |
|---|
| Glycine receptor antagonists | |
|---|
| Glutamate receptor agonists | |
|---|
| Convulsant barbiturates |
- CHEB
- Diberal (DMBB)
- McN-481
|
|---|
| Other | |
|---|
|
|---|
| Ionotropic | | GABAATooltip γ-Aminobutyric acid A receptor |
- Positive modulators (abridged; see here for a full list): α-EMTBL
- Alcohols (e.g., drinking alcohol, 2M2B)
- Anabolic steroids
- Avermectins (e.g., ivermectin)
- Barbiturates (e.g., phenobarbital)
- Benzodiazepines (e.g., diazepam)
- Bromide compounds (e.g., potassium bromide)
- Carbamates (e.g., meprobamate)
- Carbamazepine
- Chloralose
- Chlormezanone
- Clomethiazole
- Dihydroergolines (e.g., ergoloid (dihydroergotoxine))
- Etazepine
- Etifoxine
- Fenamates (e.g., mefenamic acid)
- Flavonoids (e.g., apigenin, hispidulin)
- Fluoxetine
- Flupirtine
- Imidazoles (e.g., etomidate)
- Kava constituents (e.g., kavain)
- Lanthanum
- Loreclezole
- Monastrol
- Neuroactive steroids (e.g., allopregnanolone, cholesterol, THDOC)
- Niacin
- Niacinamide
- Nonbenzodiazepines (e.g., β-carbolines (e.g., abecarnil), cyclopyrrolones (e.g., zopiclone), imidazopyridines (e.g., zolpidem), pyrazolopyrimidines (e.g., zaleplon))
- Norfluoxetine
- Petrichloral
- Phenols (e.g., propofol)
- Phenytoin
- Piperidinediones (e.g., glutethimide)
- Propanidid
- Pyrazolopyridines (e.g., etazolate)
- Quinazolinones (e.g., methaqualone)
- Retigabine (ezogabine)
- ROD-188
- Skullcap constituents (e.g., baicalin)
- Stiripentol
- Sulfonylalkanes (e.g., sulfonmethane (sulfonal))
- Topiramate
- Valerian constituents (e.g., valerenic acid)
- Volatiles/gases (e.g., chloral hydrate, chloroform, diethyl ether, paraldehyde, sevoflurane)
- Antagonists: Bicuculline
- Coriamyrtin
- Dihydrosecurinine
- Famiraprinium
- Gabazine (SR-95531)
- Hydrastine
- Hyenanchin (mellitoxin)
- Iso-THIP
- PHP-501
- Pitrazepin
- Securinine
- Sinomenine
- SR-42641
- THAZ
- Thio-4-PIOL
- Thio-THIP
- Thiocolchicoside
- Tutin
- Negative modulators: 1,3M1B
- 3M2B
- 11-Ketoprogesterone
- 17-Phenylandrostenol
- α3IA
- α5IA (LS-193,268)
- β-CCB
- β-CCE
- β-CCM
- β-CCP
- β-EMGBL
- Anabolic steroids
- Amiloride
- Anisatin
- β-Lactams (e.g., penicillins, cephalosporins, carbapenems)
- Basmisanil
- Bemegride
- Bicyclic phosphates (TBPS, TBPO, IPTBO)
- BIDN
- Bilobalide
- Bupleurotoxin
- Bupropion
- CHEB
- Chlorophenylsilatrane
- Cicutoxin
- Cloflubicyne
- Cyclothiazide
- DHEA
- DHEA-S
- Dieldrin
- (+)-DMBB
- DMCM
- DMPC
- EBOB
- Etbicyphat
- FG-7142 (ZK-31906)
- Fiproles (e.g., fipronil)
- Flavonoids (e.g., amentoflavone, oroxylin A)
- Flumazenil
- Fluoroquinolones (e.g., ciprofloxacin)
- Flurothyl
- Furosemide
- Golexanolone
- Iomazenil (123I)
- IPTBO
- Isopregnanolone (sepranolone)
- L-655,708
- Laudanosine
- Lindane
- MaxiPost
- Morphine
- Morphine-3-glucuronide
- MRK-016
- Naloxone
- Naltrexone
- Nicardipine
- Nonsteroidal antiandrogens (e.g., apalutamide, bicalutamide, enzalutamide, flutamide, nilutamide)
- Oenanthotoxin
- Pentylenetetrazol (pentetrazol)
- Phenylsilatrane
- Picrotoxin (i.e., picrotin, picrotoxinin and dihydropicrotoxinin)
- PNV-001
- Pregnenolone sulfate
- Propybicyphat
- PWZ-029
- Radequinil
- Ro 15-4513
- Ro 19-4603
- RO4882224
- RO4938581
- Sarmazenil
- SCS
- Suritozole
- TB-21007
- TBOB
- TBPS
- TCS-1105
- Terbequinil
- TETS
- Thujone
- U-93631
- Zinc
- ZK-93426
|
|---|
| GABAA-ρTooltip γ-Aminobutyric acid A-rho receptor |
- Antagonists: (S)-2-MeGABA
- (S)-4-ACPBPA
- (S)-4-ACPCA
- 2-MeTACA
- 3-APMPA
- 4-ACPAM
- 4-GBA
- cis-3-ACPBPA
- Aza-THIP
- CGP-36742 (SGS-742)
- DAVA
- Gabazine (SR-95531)
- Gaboxadol (THIP)
- I4AA
- Iso-THIP
- Isonipecotic acid
- Loreclezole
- P4MPA
- P4S
- SKF-97541
- SR-95318
- SR-95813
- THAZ
- Thio-THIP
- TPMPA
- trans-3-ACPBPA
- ZAPA
|
|---|
|
|---|
| Metabotropic | | GABABTooltip γ-Aminobutyric acid B receptor |
- Positive modulators: ADX-71441
- ASP-8062
- BHF-177
- BHFF
- rac-BHFF
- BSPP
- CGP-7930
- CGP-13501
- GS-39783
- INDV-1000
- KK-92A
- Antagonists: 2-Hydroxysaclofen
- CGP-35348
- CGP-36742
- CGP-46381
- CGP-52432
- CGP-54626
- CGP-55845
- CGP-64213
- DAVA
- Homotaurine (tramiprosate, 3-APS)
- Phaclofen
- Saclofen
- SCH-50911
- Negative modulators: Compound 14
|
|---|
|
|---|
- See also
- Receptor/signaling modulators
- GABAA receptor positive modulators
- GABA metabolism/transport modulators
|
2Mo(CO)4-from-xtal-2007-3D-balls.png)
11(EtCage)-from-xtal-1993-3D-balls.png)
RuCl(EtCage)2-from-xtal-1996-3D-balls.png)