Chloralose
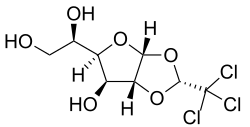
Structural formula of α-chloralose
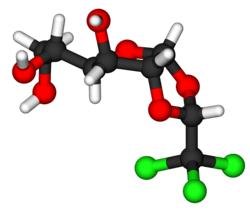
Ball-and-stick model of α-chloralose
Names
IUPAC name
1,2-O -[(1R )-2,2,2-Trichloroethane-1,1-diyl]-α-D -glucofuranose
Systematic IUPAC name
(1R )-1-[(2R ,3aR ,4S ,5R ,6aR )-2-(Trichloromethyl)-tetrahydro-2H -furo[2,3-d ][1,3]dioxol-5-yl]ethane-1,2-diol
Identifiers
CAS Number
3D model (JSmol)
Beilstein Reference
85418
ChEBI
ChEMBL
ChemSpider
ECHA InfoCard
100.036.363
EC Number
KEGG
MeSH
Chloralose
7057995 40467114 (2R ,3aR ,5R ,6R )-6-hydroxy-2-methyl-furo-5-yl 27525 (2R ,5R ,6S ,6aR )-6-hydroxy-2-methyl-furo-5-yl 16211632 (1R )-dioxol, (2R ,5R ,6S ,6aR )-6-hydroxy-2-methyl-furo-5-yl 186624 (1R )-dioxol, (3aR ,5R ,6S ,6aR )-6-hydroxy-furo-5-yl 2723807 (2R ,3aR ,5R ,6S ,6aR )-6-hydroxy-2-methyl-furo-5-yl 85991
RTECS number
UNII
InChI=1S/C8H11Cl3O6/c9-8(10,11)7-16-5-3(14)4(2(13)1-12)15-6(5)17-7/h2-7,12-14H,1H2/t2-,3+,4-,5-,6-,7-/m1/s1
Y Key: OJYGBLRPYBAHRT-IPQSZEQASA-N
Y InChI=1/C8H11Cl3O6/c9-8(10,11)7-16-5-3(14)4(2(13)1-12)15-6(5)17-7/h2-7,12-14H,1H2/t2-,3+,4-,5-,6-,7-/m1/s1
Key: OJYGBLRPYBAHRT-IPQSZEQABF
C([C@H]([C@@H]1[C@@H]([C@@H]2[C@H](O1)O[C@@H](O2)C(Cl)(Cl)Cl)O)O)O
Properties
Chemical formula
C 8 H 11 Cl 3 O 6
Molar mass
−1
Melting point
176 to 182 °C (349 to 360 °F; 449 to 455 K)
Hazards
Occupational safety and health (OHS/OSH):
Main hazards
Harmful if swallowedHarmful if inhaled
GHS labelling:
Pictograms
Signal word
Danger
Hazard statements
H301 , H332 , H336 , H410
Precautionary statements
P261 , P264 , P270 , P271 , P273 , P301+P310 , P304+P312 , P304+P340 , P312 , P321 , P330 , P391 , P403+P233 , P405 , P501
Related compounds
Related compounds
Chloral hydrate
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
Chloralose (also known as α-chloralose) is an avicide, and a rodenticide used to kill mice in temperatures below 15 °C. It is also widely used in neuroscience and veterinary medicine as an anesthetic and sedative.[ 1] urethane , it is used for long-lasting, but light anesthesia.[ 2] [ 3]
Chemically, it is a chlorinated acetal derivative of glucose .
Chloralose exerts barbiturate -like actions on synaptic transmission in the brain, including potent effects at inhibitory γ-aminobutyric acid type A receptors (GABAA R).[ 4] [ 5] A R modulator and also as a general anesthetic.[ 6]
Chloralose is often abused for its avicide properties. In the United Kingdom, protected birds of prey have been killed using the chemical.
Legal use for bird control also often causes raptor mortalities from secondary poisoning, as well as primary poisoning of non-target species from eating bait, for example, kererū pigeon in New Zealand.[ 7]
References
^ Silverman J, Muir WW (Jun 1993). "A review of laboratory animal anesthesia with chloral hydrate and chloralose". Lab Anim Sci . 43 (3): 210– 6. PMID 8355479 . ^ Vogler, George A. (2006-01-01), Suckow, Mark A.; Weisbroth, Steven H.; Franklin, Craig L. (eds.), "Chapter 19 - Anesthesia and Analgesia" , The Laboratory Rat (Second Edition) , American College of Laboratory Animal Medicine, Burlington: Academic Press, pp. 627– 664, ISBN 978-0-12-074903-4 , retrieved 2021-03-21 {{citation}}: CS1 maint: work parameter with ISBN (link)^ "Capturing problematic urban Canada geese in Reno, Nevada: Goose roundups vs. use of alpha-chloralose" . University of California. Retrieved 2025-03-26 .^ R. A. Nicoll & J. M. Wojtowicz (1980). "The effects of pentobarbital and related compounds on frog motoneurons". Brain Research . 191 (1): 225– 237. doi:10.1016/0006-8993(80)90325-x . PMID 6247012 . S2CID 21777453 . ^ K. M. Garrett & J. Gan (1998). "Enhancement of gamma-aminobutyric acidA receptor activity by alpha-chloralose". The Journal of Pharmacology and Experimental Therapeutics . 285 (2): 680– 686. doi:10.1016/S0022-3565(24)37470-1 . PMID 9580613 . ^ M. D. Krasowski & N. L. Harrison (2000). "The actions of ether, alcohol and alkane general anaesthetics on GABAA and glycine receptors and the effects of TM2 and TM3 mutations" . British Journal of Pharmacology . 129 (4): 731– 743. doi:10.1038/sj.bjp.0703087 . PMC 1571881 10683198 . ^ "Poisoned bird had enough toxin to 'kill a child' . BBC News . 2020-07-26. Retrieved 2020-07-26 .
Hypnotics/sedatives (N05C)
GABAA
Alcohols Barbiturates Benzodiazepines Carbamates Imidazoles Monoureides
Acecarbromal
Apronal (apronalide)
Bromisoval
Capuride
Carbromal Ectylurea Neurosteroids Nonbenzodiazepines Phenols
Cipepofol (ciprofol)
Fospropofol
Propofol Propofol hemisuccinate Piperidinediones Quinazolinones
Afloqualone
Cloroqualone
Diproqualone
Etaqualone
Mebroqualone
Mecloqualone
Methaqualone Methylmethaqualone
Nitromethaqualone
SL-164 Others
GABAB H1
Antihistamines Antidepressants
Serotonin antagonists and reuptake inhibitors
Tricyclic antidepressants
Tetracyclic antidepressants
Antipsychotics
Typical antipsychotics
Atypical antipsychotics
α2 -Adrenergic 5-HT2A
Antidepressants
Trazodone Tricyclic antidepressants
Tetracyclic antidepressants
Antipsychotics
Typical antipsychotics
Atypical antipsychotics
Others
Melatonin Orexin
Daridorexant
Lemborexant
Suvorexant α2 δ VDCC Others
General anesthetics (N01A)
Inhalational Injection
# WHO-EM‡ Withdrawn from marketClinical trials:
† Phase III§ Never to phase III
Pest control: Rodenticides
Anticoagulants /
Convulsants Calciferols Inorganic compounds Organochlorine Organophosphorus Carbamates Others
GABA A receptor positive modulators
Alcohols Barbiturates Benzodiazepines Carbamates
Carisbamate
Carisoprodol Clocental
Cyclarbamate
Difebarbamate
Emylcamate
Ethinamate Febarbamate
Felbamate Hexapropymate
Hydroxyphenamate
Lorbamate
Mebutamate
Meprobamate Nisobamate
Pentabamate
Phenprobamate
Procymate
Styramate
Tetrabamate
Tybamate Flavonoids Imidazoles
Etomidate Metomidate
Methoxyetomidate
Propoxate
Isopropoxate
Butomidate
Iso-butomidate
Sec-butomidate
CF2-Etomidate
CF3-Etomidate
CF3-Propoxate
Flutomidate
2,6-Dichloro-3-fluoroetomidate Kava constituents
10-Methoxyyangonin
11-Methoxyyangonin
11-Hydroxyyangonin
Desmethoxyyangonin
11-Methoxy-12-hydroxydehydrokavain
7,8-Dihydroyangonin
Kavain
5-Hydroxykavain
5,6-Dihydroyangonin
7,8-Dihydrokavain
5,6,7,8-Tetrahydroyangonin
5,6-Dehydromethysticin
Methysticin 7,8-Dihydromethysticin
Yangonin Monoureides
Acecarbromal
Apronal (apronalide)
Bromisoval
Carbromal Capuride
Ectylurea Neuroactive steroids Nonbenzodiazepines
Imidazopyridines : AlpidemDS-1
Necopidem
Saripidem
Zolpidem Pyrazolopyrimidines : DivaplonFasiplon
Indiplon
Lorediplon
Ocinaplon
Panadiplon
Taniplon
Zaleplon Others : AdipiplonAXS-17 (BAER-101, AZD-7325)
CGS-8216
CGS-9896
CGS-13767
CGS-20625
CL-218,872
CP-615,003
CTP-354
ELB-139
GBLD-345
Imepitoin
JM-1232
L-838,417
Lirequinil (Ro41-3696)
Miltirone (rosmariquinone)
NS-2664
NS-2710
NS-11394
Pipequaline
ROD-188
RWJ-51204
SB-205,384
SX-3228
TGSC01AA
TP-003
TPA-023
TP-13
U-89843A
U-90042
Viqualine
Y-23684 Phenols Piperidinediones Pyrazolopyridines
Cartazolate
Etazolate
ICI-190,622
Tracazolate Quinazolinones
Afloqualone
Cloroqualone
Diproqualone
Etaqualone
Mebroqualone
Mecloqualone
Methaqualone Methylmethaqualone
Nitromethaqualone
SL-164 Volatiles/gases Others/unsorted
3-Hydroxybutanal α-EMTBL
AA-29504
Alogabat
Avermectins (e.g., ivermectin )Bromide compounds (e.g., lithium bromide , potassium bromide , sodium bromide )
Carbamazepine Chloralose Chlormezanone Clomethiazole
Darigabat
DEABL
Deuterated etifoxine
Dihydroergolines (e.g., dihydroergocryptine, dihydroergosine, dihydroergotamine , ergoloid (dihydroergotoxine) )
DS2
Efavirenz Etazepine
Etifoxine
Fenamates (e.g., flufenamic acid , mefenamic acid , niflumic acid , tolfenamic acid )Fluoxetine Flupirtine
Hopantenic acid
KRM-II-81
Lanthanum Lavender oil
Lignans (e.g., 4-O-methylhonokiol, honokiol , magnolol , obovatol)
Loreclezole
Menthyl isovalerate (validolum) Monastrol Nicotinic acid Nicotinamide Org 25,435
Phenytoin Propanidid
Retigabine (ezogabine)
Safranal Seproxetine
Stiripentol
Sulfonylalkanes (e.g., sulfonmethane (sulfonal), tetronal, trional)
Terpenoids (e.g., borneol )
Topiramate Valerian constituents (e.g., isovaleric acid , isovaleramide, valerenic acid, valerenol) Unsorted benzodiazepine site positive modulators: α-Pinene MRK-409 (MK-0343)
TCS-1105
TCS-1205 See also: Receptor/signaling modulators • GABA receptor modulators • GABA metabolism/transport modulators