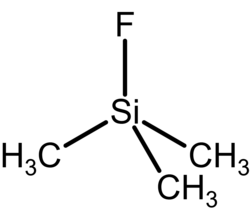
Trimethylsilyl fluoride
 | |
| Names | |
|---|---|
| IUPAC name
Fluoro(trimethyl)silane[1]
| |
| Other names | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.006.362 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
(CH3)3SiF |
| Molar mass | 92.188 g·mol−1 |
| Appearance | Colorless gas[3] |
| Density | 0.793 g/cm3 at 0 °C |
| Melting point | −74 °C (−101 °F; 199 K)[3] |
| Boiling point | 16 °C (61 °F; 289 K)[3] |
Solubility in water
|
Reacts[4] |
| Solubility | Soluble in most organic solvents, except the protic ones with which it reacts.[4] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Skin burns and serious eye damage |
| GHS labelling: | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H224, H280, H315, H319, H335 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P261, P264, P264+P265, P271, P280, P302+P352, P303+P361+P353, P304+P340, P305+P351+P338, P319, P321, P332+P317, P337+P317, P362+P364, P370+P378, P403+P233, P403+P235, P405, P410+P403, P501 |
| Flash point | -30 °C[3] |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Trimethylsilyl fluoride is an organosilicon compound with the formula (CH3)3SiF. It is a colorless gas.
Synthesis
Trimethylsilyl fluoride can be synthesized from trimethylsilyl chloride and potassium fluoride in the presence of a phase-transfer catalyst and a few drops of water.[5] It can also be synthesized by the reaction of trimethylsilyl trifluoromethanesulfonate with potassium fluoride in dimethylformamide containing 18-crown-6 ether.[6] It can be synthesized from trimethylsilyl chloride by treatment with hydrogen fluoride.
- (CH3)3SiCl + HF → (CH3)3SiF + HCl
It can also be generated in situ by reaction of ethyl trimethylsilylacetate and tetra-n-butylammonium fluoride.[4]
Uses
Trimethylsilyl fluoride is used as a protecting group during synthesis of certain chemicals and during certain reactions (e.g. silylation). Its applications are in pharmaceutical industry and organic synthesis.[7]
Reactions
Trimethylsilyl fluoride reacts with norbornyllithium to give trimethylsilylnorbornane.[8] Trimethylsilyl fluoride can silylate ketones, alcohols, and terminal alkynes.[4]
Safety
Trimethylsilyl fluoride is a dermatotoxin. Cause skin, eye and respiratory system irritation. It causes skin burns and serious eye damage. It can cause chemical pneumonitis.[1] It is extremely flammable and its vapors can form explosive mixtures with air.[3]
References
- ^ a b c d e "Fluorotrimethylsilane". pubchem.ncbi.nlm.nih.gov. Retrieved 25 February 2026.
- ^ Wilson, W. W.; Haiges, R.; Christe, K. O. (1 August 2023). "High resolution NMR spectra of fluorotrimethylsilane". Journal of Fluorine Chemistry. 270 110166. doi:10.1016/j.jfluchem.2023.110166. Retrieved 25 February 2026 – via ScienceDirect.
- ^ a b c d e https://www.sigmaaldrich.com/GB/en/sds/aldrich/364533
- ^ a b c d "Trimethylfluorosilane | 420-56-4". ChemicalBook. Retrieved 25 February 2026.
- ^ Dehmlow, E. V.; Fastabend, U.; Keßler, M. (25 February 1988). "A One-Pot Synthesis of Trimethylsilyl Fluoride". Synthesis. 1988 (12): 996–997. doi:10.1055/s-1988-27783. Retrieved 25 February 2026 – via www.thieme-connect.com.
- ^ Della, Ernest W.; Tsanaktsidis, John (25 May 1988). "A Convenient Synthesis of Trimethylsilyl Fluoride". Synthesis. 5 (5): 407. doi:10.1055/s-1988-27596. Retrieved 25 February 2026 – via www.thieme-connect.com.
- ^ Bull, Chemical. "Trimethylsilyl Fluoride | 420-56-4 | Chemical Bull Pvt. Ltd". www.chemicalbull.com. Retrieved 25 February 2026.
- ^ Della, Ernest W.; Tsanaktsidis, John. (1 May 1988). "Synthesis of some bridgehead (trimethylsilyl)polycycloalkanes. Silicon-29 NMR chemical shifts and silicon-29-carbon-13 coupling constants". Organometallics. 7 (5): 1178–1182. doi:10.1021/om00095a025. Retrieved 25 February 2026 – via ACS Publications.