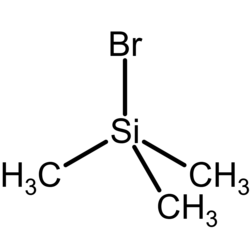
Trimethylsilyl bromide
 | |
| Names | |
|---|---|
| IUPAC name
Bromo(trimethyl)silane[1]
| |
| Other names | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.018.793 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
(CH3)3SiBr |
| Molar mass | 153.094 g·mol−1 |
| Appearance | Colorless liquid[2] |
| Density | 1.16 g/cm3[3] |
| Melting point | −43 °C (−45 °F; 230 K)[4] |
| Boiling point | 79 °C (174 °F; 352 K)[3] |
Solubility in water
|
Reacts |
| Solubility | Soluble in carbon tetrachloride, chloroform, dichloromethane, 1,2-dichloroethane, toluene, hexanes[2] Reactive with alcohols, ethers and esters.[2] |
Refractive index (nD)
|
1.4240[2] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Causes severe skin burns and eye damage |
| GHS labelling: | |
Pictograms
|
   
|
Signal word
|
Danger |
Hazard statements
|
H225, H226, H314, H410 |
Precautionary statements
|
P210, P233, P240, P241, P242, P243, P260, P264, P280, P301+P330+P331, P302+P361+P354, P303+P361+P353, P304+P340, P305+P354+P338, P316, P321, P363, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 
3
3
2 |
| Flash point | 32 °C[3] |
| Related compounds | |
Related compounds
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Trimethylsilyl bromide is an organosilicon compound with formula (CH3)3SiBr. It is a colorless liquid.
Synthesis
Trimethylsilyl bromide can be synthesized by halogen exchange between trimethylsilyl chloride and either magnesium bromide in diethyl ether or sodium bromide in acetonitrile.[2]
- 2 (CH3)3SiCl + MgBr2 → 2 (CH3)3SiBr + MgCl2
- (CH3)3SiCl + NaBr → (CH3)3SiBr + NaCl
It can also be synthesized by reaction between hexamethyldisilane and bromine in benzene, with no byproducts.[2]
- (CH3)3Si−Si(CH3)3 + Br2 → 2 (CH3)3SiBr
Trimethylsilyl bromide can be prepared in a laboratory scale from trimethylsilyl-4-bromo-2-alkenoates in almost quantitative yield.[5]
It can also be synthesized by reaction of hexamethyldisiloxane and aluminium bromide.[2]
Characteristics
Trimethylsilyl bromide is a colorless liquid very sensitive to light, air and moisture. It fumes in air upon hydrolysis, producing hydrobromic acid. Upon prolonged storage it becomes yellow to orange because of releasing of bromine.[2] Reacts violently with water.[3]
Uses
Trimethylsilyl bromide is used as a mild and selective reagent for cleavage of lactones, epoxides, acetals, phosphonate esters and certain ethers. It is effective reagent for formation of silyl enol ethers.[2] Trimethylsilyl bromide is as a mild, stereoselective anomeric brominating agent. Stereoselective bromination of anomeric glycosyl acetates is achieved with trimethylsilyl bromide under mild conditions and in the presence of various protecting groups commonly employed in carbohydrate chemistry.[6]
References
- ^ a b c "Bromotrimethylsilane". pubchem.ncbi.nlm.nih.gov.
- ^ a b c d e f g h i Martinelli, Michael J. (2001-04-15), "Bromotrimethylsilane", in John Wiley & Sons, Ltd (ed.), Encyclopedia of Reagents for Organic Synthesis, Chichester: John Wiley & Sons, Ltd, doi:10.1002/047084289X.rb327, ISBN 978-0-471-93623-7, retrieved 2026-02-25
- ^ a b c d "Bromotrimethylsilane". sigmaaldrich.com. Retrieved February 24, 2026.
- ^ "Bromotrimethylsilane | 2857-97-8". ChemicalBook. Retrieved 2026-02-25.
- ^ Bellassoued, M.; Borgi, A. El; Gaudemar, M. (September 1985). "A New Convenient Laboratory Synthesis of Trimethylsilyl Bromide". Synthetic Communications. 15 (11): 973–975. doi:10.1080/00397918508076828. ISSN 0039-7911.
- ^ Gillard, John W; Israel, Mervyn (1981-01-01). "Trimethylsilyl bromide as a mild, stereoselective anomeric brominating agent". Tetrahedron Letters. 22 (6): 513–516. doi:10.1016/S0040-4039(01)90142-X. ISSN 0040-4039.