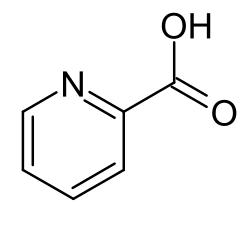
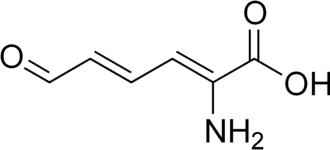
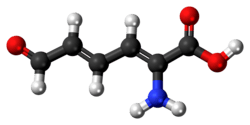
2-Aminomuconic semialdehyde
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
(2Z,4E)-2-Amino-6-oxohexa-2,4-dienoic acid | |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
PubChem CID
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C6H7NO3 |
| Molar mass | 141.12 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
2-Aminomuconic semialdehyde is a metabolite of tryptophan which is an unstable open-chain biochemical precursor of picolinic acid.[1][2][3]
Biochemistry
2-Aminomuconic semialdehyde is an intermediate in the kynurenine pathway of tryptophan metabolism, which leads to picolinic acid or quinolinic acid. The enzyme aminocarboxymuconate-semialdehyde decarboxylase produces it from 2-amino-3-carboxymuconic semialdehyde, a compound which would form quinolinic acid in the absence of that enzyme.[3]
2-Aminomuconic semialdehyde itself is chemically unstable and spontaneously ring-closes to picolinic acid, with loss of water.[3][4]
See also
- Muconic acid
- Ommochrome
References
- ^ He, Z.; Spain, J. (1999-08-01). "Preparation of 2-aminomuconate from 2-aminophenol by coupled enzymatic dioxygenation and dehydrogenation reactions". Journal of Industrial Microbiology & Biotechnology. 23 (2): 138–142. doi:10.1038/sj.jim.2900705. ISSN 1476-5535. PMID 10510494. S2CID 1091252.
- ^ Zeng, Ting; Liang, Yanshan; Chen, Jinyao; Cao, Guodong; Yang, Zhu; Zhao, Xingchen; Tian, Jinglin; Xin, Xiong; Lei, Bo; Cai, Zongwei (2021-09-01). "Urinary metabolic characterization with nephrotoxicity for residents under cadmium exposure". Environment International. 154 106646. Bibcode:2021EnInt.15406646Z. doi:10.1016/j.envint.2021.106646. ISSN 0160-4120. PMID 34049269.
- ^ a b c Savitz, J (25 January 2020). "The kynurenine pathway: a finger in every pie". Molecular Psychiatry. 25 (1): 131–147. doi:10.1038/s41380-019-0414-4. PMC 6790159. PMID 30980044.
- ^ Ichiyama, Arata; Nakamura, Shigenobu; Kawai, Hitoshi; Honjo, Tasuku; Nishizuka, Yasutomi; Hayaishi, Osamu; Senoh, Siro (1965). "Studies on the Metabolism of the Benzene Ring of Tryptophan in Mammalian Tissues". Journal of Biological Chemistry. 240 (2): 740–749. doi:10.1016/S0021-9258(17)45238-0.