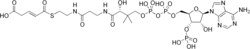Glutaconyl-CoA
| Names | |
|---|---|
| IUPAC name
(3E)-5-[(2-{3-[(2R)-4-{[1,3-Dihydroxy-1,3-dioxo-3-(3′-O-phosphonoadenosin-5′-O-yl)-1λ5,3λ5-diphosphoxan-1-yl]oxy}-3,3-dimethylbutanamido]propanamido}ethyl)sulfanyl]-5-oxopent-3-enoic acid
| |
| Systematic IUPAC name
(9R,20E)-1-[(2R,3S,4R,5R)-5-(6-Amino-9H-purin-9-yl)-4-hydroxy-3-(phosphonooxy)oxolan-2-yl]-3,5,9-trihydroxy-8,8-dimethyl-3,5,10,14,19-pentaoxo-2,4,6-trioxa-18-thia-11,15-diaza-3λ5,5λ5-diphosphatricos-20-en-23-oic acid | |
| Other names
Glutaconyl-coenzyme A
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| MeSH | glutaconyl-coenzyme+A |
PubChem CID
|
|
| UNII | |
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C26H40N7O19P3S |
| Molar mass | 879.62 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Glutaconyl-CoA is an intermediate in the metabolism of lysine.[1] It is an organic compound containing a coenzyme substructure, which classifies it as a fatty ester lipid molecule. Being a lipid makes the molecule hydrophobic, which makes it insoluble in water. The molecule has a molecular formula of C26H40N7O19P3S, and a molecular weight 879.62 grams per mole.[2]
Glutaconyl-CoA is postulated to be the main toxin in glutaric aciduria type 1.[3] In certain fermentative bacteria, glutaconyl-CoA decarboxylation is catalyzed by a Na+-dependent decarboxylase (EC 7.2.4.5) and is coupled with Na+ ion translocation, which creates a sodium-motive force as an alternate energy source for these organisms.[4]
See also
- Glutaconate CoA-transferase
- Glutaconyl-CoA decarboxylase
References
- ^ Trefely, Sophie; Lovell, Claudia D.; Snyder, Nathaniel W.; Wellen, Kathryn E. (2020). "Compartmentalised acyl-CoA metabolism and roles in chromatin regulation". Molecular Metabolism. 38 100941. doi:10.1016/j.molmet.2020.01.005. PMC 7300382. PMID 32199817.
Glutaryl-CoA is a product of tryptophan and lysine catabolism preceding crotonoyl-CoA production in the mitochondria
- ^ "Human Metabolome Database: Showing metabocard for Glutaconyl-CoA (HMDB0001290)". hmdb.ca. Retrieved 2022-10-18.
- ^ Lehnert, Willy; Sass, Jörn Oliver (January 2005). "Glutaconyl-CoA is the main toxic agent in glutaryl-CoA dehydrogenase deficiency (glutaric aciduria type I)". Medical Hypotheses. 65 (2): 330–333. doi:10.1016/j.mehy.2005.02.021. PMID 15922108.
- ^ Kress, Daniel; Brügel, Daniela; Schall, Iris; Linder, Dietmar; Buckel, Wolfgang; Essen, Lars-Oliver (October 2009). "An Asymmetric Model for Na+-translocating Glutaconyl-CoA Decarboxylases". Journal of Biological Chemistry. 284 (41): 28401–28409. doi:10.1074/jbc.m109.037762. PMC 2788889. PMID 19654317.