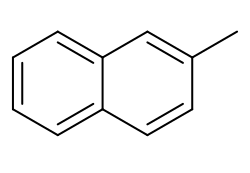
2-Methylnaphthalene
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2-Methylnaphthalene | |
| Other names
β-methylnaphthalene
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.001.890 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C11H10 |
| Molar mass | 142.201 g·mol−1 |
| Appearance | Waxy white solid |
| Melting point | 35 °C (95 °F; 308 K) |
| Boiling point | 241.1 °C (466.0 °F; 514.2 K)[1] |
Magnetic susceptibility (χ)
|
−102.6·10−6 cm3/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
2-Methylnaphthalene is a simple polycyclic aromatic hydrocarbon (PAH). It is generally isolated from coal tar, of which it is a minor component.[2]
Uses
The quinone derivative, Menadione, can be formed by the oxidation of 2-methylnaphthalene and finds use as a synthetic form of vitamin K.[3]
Astrochemistry
According to NASA, over 20% of the carbon in the universe may be associated with PAHs, possible starting materials for the formation of life.[4] PAHs seem to have been formed shortly after the Big Bang, are abundant in the universe,[5][6][7] and are associated with new stars and exoplanets.[4]
See also
References
- ^ Feldman, Julian; Orchin, Milton (December 1952). "Separation of 1- and 2-Methylnaphthalenes by Azeotropic Distillation". Industrial & Engineering Chemistry. 44 (12): 2909–2914. doi:10.1021/ie50516a041.
- ^ Gerd Collin; Hartmut Höke; Helmut Greim (2003). "Naphthalene and Hydronaphthalenes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. ISBN 978-3-527-30673-2..
- ^ de Souza, Acácio S; Ribeiro, Ruan Carlos B; Costa, Dora C S; Pauli, Fernanda P; Pinho, David R; de Moraes, Matheus G; da Silva, Fernando de C; Forezi, Luana da S M; Ferreira, Vitor F (11 April 2022). "Menadione: a platform and a target to valuable compounds synthesis". Beilstein Journal of Organic Chemistry. 18: 381–419. doi:10.3762/bjoc.18.43. PMC 9039524.
- ^ a b Hoover, Rachel (February 21, 2014). "Need to Track Organic Nano-Particles Across the Universe? NASA's Got an App for That". NASA. Retrieved February 22, 2014.
- ^ Carey, Bjorn (October 18, 2005). "Life's Building Blocks 'Abundant in Space'". Space.com. Retrieved March 3, 2014.
- ^ Hudgins, Douglas M.; Bauschlicher Jr, Charles W.; Allamandola, L. J. (October 10, 2005). "Variations in the Peak Position of the 6.2 μm Interstellar Emission Feature: A Tracer of N in the Interstellar Polycyclic Aromatic Hydrocarbon Population". Astrophysical Journal. 632 (1): 316–332. Bibcode:2005ApJ...632..316H. doi:10.1086/432495.
- ^ Allamandola, Louis; et al. (April 13, 2011). "Cosmic Distribution of Chemical Complexity". NASA. Archived from the original on February 27, 2014. Retrieved March 3, 2014.