Phenanthrene
Phenanthrene
Names
Preferred IUPAC name
Identifiers
CAS Number
3D model (JSmol)
Beilstein Reference
1905428
ChEBI
ChemSpider
ECHA InfoCard
100.001.437
EC Number
Gmelin Reference
28699
KEGG
MeSH
C031181
UNII
InChI=1S/C14H10/c1-3-7-13-11(5-1)9-10-12-6-2-4-8-14(12)13/h1-10H
N Key: YNPNZTXNASCQKK-UHFFFAOYSA-N
N InChI=1/C14H10/c1-3-7-13-11(5-1)9-10-12-6-2-4-8-14(12)13/h1-10H
Key: YNPNZTXNASCQKK-UHFFFAOYAC
C1=CC=C2C(=C1)C=CC3=CC=CC=C32
Properties
Chemical formula
C 14 H 10
Molar mass
−1
Appearance
Colorless solid
Density
1.18 g/cm3 [ 1]
Melting point
101 °C (214 °F; 374 K)[ 1]
Boiling point
332 °C (630 °F; 605 K)[ 1]
Solubility in water
1.6 mg/L[ 1]
Magnetic susceptibility (χ)
−127.9·10−6 cm3 /mol
Hazards
NFPA 704 (fire diamond)
Flash point
171 °C (340 °F; 444 K)[ 1]
Structure
Point group
C2v [ 2]
Dipole moment
0 D
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
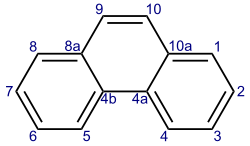
Phenanthrene is a polycyclic aromatic hydrocarbon (PAH) with formula C14 H10 , consisting of three fused benzene rings. It is a colorless, crystal-like solid, but can also appear yellow. Phenanthrene is used to make dyes, plastics, pesticides, explosives, and drugs. It has also been used to make bile acids, cholesterol and steroids.[ 3]
Phenanthrene occurs naturally and also is a man-made chemical. Commonly, humans are exposed to phenanthrene through inhalation of cigarette smoke, but there are many routes of exposure. Animal studies have shown that phenanthrene is a potential carcinogen.[ 3] [ 4]
Phenanthrene's three fused rings are angled as in the phenacenes, rather than straight as in the acenes. The compounds with a phenanthrene skeleton but with nitrogen atoms in place of CH sites are known as phenanthrolines .
History and etymology
Phenanthrene was discovered in coal tar in 1872 independently by Carl Graebe (article manuscript received on November 1st[ 5] [ 6] [ 7] corresponding quinone and then to diphenic acid , and soon Graebe confirmed it by a synthesis from stilbene .[ 8]
Prior to February 1873 Fittig sent a letter to Graebe where he proposed to name the hydrocarbon phenanthrene (German: Phenanthren ) in order to account for its similarity to biphenyl and anthracene , which was swiftly adopted.[ 9]
Physical properties
Phenanthrene is nearly insoluble in water but is soluble in most low-polarity organic solvents such as toluene , carbon tetrachloride , ether , chloroform , acetic acid and benzene .
Phenanthrene is fluorescent under ultraviolet light, exhibiting a large Stoke shift.[ 10]
Chemistry
Reactions of phenanthrene typically occur at the 9 and 10 positions, including:
Productions
Phenanthrene is extracted from coal tar, of which it comprises 5% by weight.[ 16]
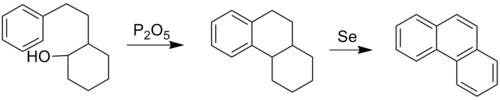
In principle it could be obtained by chemical synthesis. The Bardhan–Sengupta phenanthrene synthesis is a classic way to make phenanthrenes.[ 17]
This process involves electrophilic aromatic substitution using a tethered cyclohexanol group using diphosphorus pentoxide , which closes the central ring onto an existing aromatic ring. Dehydrogenation using selenium aromatizes the other rings into aromatic ones as well. The aromatization of six-membered rings produces H2 Se .
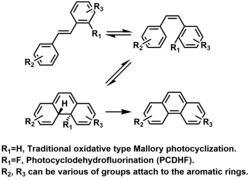
Phenanthrene can also be obtained photochemically from certain diarylethenes (Mallory reaction):
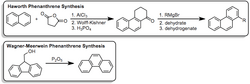
Other synthesis routes include the Haworth reaction and the Wagner-Meerwein-type ring-expansion, as depicted below:
Commercially phenanthrene is not synthesized but extracted from the byproducts of coal coking, since it makes around 4–6% of coke oven coal tar.[ 18]
Natural occurrences of the phenanthrene derivatives
Structure of morphinan, which features a partially reduced phenanthroline core Morphinan is the chemical structure found in several psychoactive drugs, consisting of opiate analgesics, cough suppressants, and dissociative hallucinogens, among others. Examples morphine , codeine , and dextromethorphan (DXM).[ 16]
Ravatite is a natural mineral consisting of phenanthrene.[ 19]
In plants
Phenanthrene derivatives occur in plants as phenanthrenoids . They have been reported from flowering plants, mainly in the family Orchidaceae, and a few in the families Dioscoreaceae, Combretaceae and Betulaceae, as well as in the lower plant class Marchantiophyta (liverworts).[ 20]
See also
References
^ a b c d e Record of CAS RN 85-01-8 in the GESTIS Substance Database of the Institute for Occupational Safety and Health^ Peter Atkins, J. D. P., Atkins' Physical Chemistry. Oxford: 2010. P. 443.
^ a b "Phenanthrene Fact Sheet" (PDF) . archive.epa.gov . U.S. Environmental Protection Agency. Retrieved 19 July 2019 .^ "Phenanthrene" . Sigma-Alrdich .^ Graebe, C. (1872). "Ueber einen neuen dem Anthracen isomeren Kohlenwasserstoff" Berichte der deutschen chemischen Gesellschaft . 5 (2): 861– 863. doi:10.1002/cber.18720050279 . ISSN 0365-9496 . ^ Ostermayer, E.; Fittig, R. (1872). "Ueber einen neuen Kohlenwasserstoff aus dem Steinkohlentheer" . Berichte der deutschen chemischen Gesellschaft . 5 (2): 933– 937. doi:10.1002/cber.187200502100 . ISSN 0365-9496 . ^ Ostermayer, Eugen (1872). Ueber einen neuen Kohlenwasserstoff im Steinkohlentheeröl: Inaugural-Dissertation ^ Graebe, C. (1873). "Ueber Synthese des Phenanthrens" . Berichte der deutschen chemischen Gesellschaft . 6 (1): 125– 127. doi:10.1002/cber.18730060147 . ISSN 0365-9496 . ^ Graebe, C. (1873). "Ueber das Verhalten der Chinone beim Erhitzen mit Natronkalk" Berichte der deutschen chemischen Gesellschaft . 6 (1): 63– 66. doi:10.1002/cber.18730060124 . ISSN 0365-9496 . ^ "Spectrum [Phenanthrene] | AAT Bioquest" . www.aatbio.com . Retrieved 2024-07-30 .^ Organic Syntheses, Coll. Vol. 4, p. 757 (1963); Vol. 34, p. 76 (1954).
^ Organic Syntheses, Coll. Vol. 4, p. 313 (1963); Vol. 34, p. 31 (1954).
^ Organic Syntheses, Coll. Vol. 3, p. 134 (1955); Vol. 28, p. 19 (1948).
^ Organic Syntheses, Coll. Vol. 2, p. 482 (1943); Vol. 16, p. 63 (1936).
^ Organic Syntheses, Coll. Vol. 5, p. 489 (1973); Vol. 41, p. 41 (1961).
^ a b Schmidt, Roland; Griesbaum, Karl; Behr, Arno; Biedenkapp, Dieter; Voges, Heinz-Werner; Garbe, Dorothea; Paetz, Christian; Collin, Gerd; Mayer, Dieter; Höke, Hartmut (2014). "Hydrocarbons". Ullmann's Encyclopedia of Industrial Chemistry . pp. 1– 74. doi:10.1002/14356007.a13_227.pub3 . ISBN 978-3-527-30673-2 . ^ "Bardhan Sengupta Synthesis". Comprehensive Organic Name Reactions and Reagents . Vol. 49. 2010. pp. 215– 219. doi:10.1002/9780470638859.conrr049 . ISBN 978-0-470-63885-9 . ^ Ma, Zhi-Hao; Wei, Xian-Yong; Liu, Guang-Hui; Liu, Fang-Jing; Zong, Zhi-Min (2021-05-15). "Value-added utilization of high-temperature coal tar: A review" Fuel . 292 119954. doi:10.1016/j.fuel.2020.119954 . ISSN 0016-2361 . ^ Ravatite Mineral Data ^ Kovács, Adriána; Vasas, Andrea; Hohmann, Judit (2008). "Natural phenanthrenes and their biological activity". Phytochemistry . 69 (5): 1084– 1110. Bibcode:2008PChem..69.1084K . doi:10.1016/j.phytochem.2007.12.005 . PMID 18243254 .
External links
Hydrocarbons
Saturated
Alkanesn 2n + 2
Linear alkanes Branched alkanes
Cycloalkanes Alkylcycloalkanes Bicycloalkanes
Housane (bicyclo[2.1.0]pentane)
Norbornane (bicyclo[2.2.1]heptane)Decalin (bicyclo[4.4.0]decane) Polycycloalkanes
Adamantane Diamondoid
Perhydrophenanthrene
Sterane
Cubane Prismane
Dodecahedrane
Basketane
Churchane
Pagodane
Twistane Other
Unsaturated
Alkenesn 2n
Linear alkenes Branched alkenes
Alkynesn 2n − 2
Linear alkynes Branched alkynes
Isopentyne
Isohexyne
Isoheptyne
Isooctyne
Isononyne
Isodecyne
Cycloalkenes Alkylcycloalkenes
Methylcyclopropene
Methylcyclobutene
Methylcyclopentene
Methylcyclohexene Isopropylcyclohexene
Bicycloalkenes Cycloalkynes Dienes Other
Aromatic
PAHs
Alkylbenzenes C2-Benzenes
C3-Benzenes
C4-Benzenes
Other
Vinylbenzenes Other
Other
Polycyclic aromatic hydrocarbons
2 rings 3 rings 4 rings 5 rings 6 rings 7+ rings
Coronene Circumcoronene
Dicoronylene
Diindenoperylene
Heptacene
Hexabenzocoronene (Hexa-cata-)
Kekulene
Octacene
Ovalene
Rubicene Rubrene Sumanene
Superphenalene
Trinaphthylene
Truxene General classes
Acene
Circulene
Cyclacene
Helicene
Phenacene
Topics in organic reactions
Addition reaction
Elimination reaction
Polymerization
Reagents
Rearrangement reaction
Redox reaction
Regioselectivity
Stereoselectivity
Stereospecificity
Substitution reaction
A value
Alpha effect
Annulene
Anomeric effect
Antiaromaticity
Aromatic ring current
Aromaticity
Baird's rule
Baker–Nathan effect
Baldwin's rules
Bema Hapothle
Beta-silicon effect
Bicycloaromaticity
Bredt's rule
Bürgi–Dunitz angle
Catalytic resonance theory
Charge remote fragmentation
Charge-transfer complex
Clar's rule
Conformational isomerism
Conjugated system
Conrotatory and disrotatory
Curtin–Hammett principle
Dynamic binding (chemistry)
Edwards equation
Effective molarity
Electromeric effect
Electron-rich
Electron-withdrawing group
Electronic effect
Electrophile
Evelyn effect
Flippin–Lodge angle
Free-energy relationship
Grunwald–Winstein equation
Hammett acidity function
Hammett equation
George S. Hammond
Hammond's postulate
Homoaromaticity
Hückel's rule
Hyperconjugation
Inductive effect
Kinetic isotope effect
LFER solvent coefficients (data page)
Marcus theory
Markovnikov's rule
Möbius aromaticity
Möbius–Hückel concept
More O'Ferrall–Jencks plot
Negative hyperconjugation
Neighbouring group participation
2-Norbornyl cation
Nucleophile
Kennedy J. P. Orton
Passive binding
Phosphaethynolate
Polar effect
Polyfluorene
Ring strain
Σ-aromaticity
Spherical aromaticity
Spiroaromaticity
Steric effects
Superaromaticity
Swain–Lupton equation
Taft equation
Thorpe–Ingold effect
Vinylogy
Walsh diagram
Woodward–Hoffmann rules
Woodward's rules
Y-aromaticity
Yukawa–Tsuno equation
Zaitsev's rule
Σ-bishomoaromaticity List of organic reactions
Carbon-carbon Homologation reactions
Arndt–Eistert reaction
Hooker reaction
Kiliani–Fischer synthesis
Kowalski ester homologation
Methoxymethylenetriphenylphosphorane
Seyferth–Gilbert homologation
Wittig reaction Olefination reactions
Bamford–Stevens reaction
Barton–Kellogg reaction
Boord olefin synthesis
Chugaev elimination
Cope reaction
Corey–Winter olefin synthesis
Dehydrohalogenation
Elimination reaction
Grieco elimination
Hofmann elimination
Horner–Wadsworth–Emmons reaction
Hydrazone iodination
Julia olefination
Julia–Kocienski olefination
Kauffmann olefination
McMurry reaction
Peterson olefination
Ramberg–Bäcklund reaction
Shapiro reaction
Takai olefination
Wittig reaction
Carbon-heteroatom
Azo coupling
Bartoli indole synthesis
Boudouard reaction
Cadogan–Sundberg indole synthesis
Diazonium compound
Esterification
Grignard reagent
Haloform reaction
Hegedus indole synthesis
Hurd–Mori 1,2,3-thiadiazole synthesis
Kharasch–Sosnovsky reaction
Knorr pyrrole synthesis
Leimgruber–Batcho indole synthesis
Mukaiyama hydration
Nenitzescu indole synthesis
Oxymercuration reaction
Reed reaction
Schotten–Baumann reaction
Ullmann condensation
Williamson ether synthesis
Yamaguchi esterification Degradation
Barbier–Wieland degradation
Bergmann degradation
Edman degradation
Emde degradation
Gallagher–Hollander degradation
Hofmann rearrangement
Hooker reaction
Isosaccharinic acid
Marker degradation
Ruff degradation
Strecker degradation
Von Braun amide degradation
Weerman degradation
Wohl degradation Organic redox
Acyloin condensation
Adkins–Peterson reaction
Akabori amino-acid reaction
Alcohol oxidation
Algar–Flynn–Oyamada reaction
Amide reduction
Andrussow process
Angeli–Rimini reaction
Aromatization
Autoxidation
Baeyer–Villiger oxidation
Barton–McCombie deoxygenation
Bechamp reduction
Benkeser reaction
Bergmann degradation
Birch reduction
Bohn–Schmidt reaction
Bosch reaction
Bouveault–Blanc reduction
Boyland–Sims oxidation
Cannizzaro reaction
Carbonyl reduction
Clemmensen reduction
Collins oxidation
Corey–Itsuno reduction
Corey–Kim oxidation
Corey–Winter olefin synthesis
Criegee oxidation
Dakin oxidation
Davis oxidation
Deoxygenation
Dess–Martin oxidation
DNA oxidation
Elbs persulfate oxidation
Emde degradation
Eschweiler–Clarke reaction
Étard reaction
Fischer–Tropsch process
Fleming–Tamao oxidation
Fukuyama reduction
Ganem oxidation
Glycol cleavage
Griesbaum coozonolysis
Grundmann aldehyde synthesis
Haloform reaction
Hydrogenation
Hydrogenolysis
Hydroxylation
Jones oxidation
Kiliani–Fischer synthesis
Kolbe electrolysis
Kornblum oxidation
Kornblum–DeLaMare rearrangement
Leuckart reaction
Ley oxidation Lindgren oxidation
Lipid peroxidation
Lombardo methylenation
Luche reduction
Markó–Lam deoxygenation
McFadyen–Stevens reaction
Meerwein–Ponndorf–Verley reduction
Methionine sulfoxide Miyaura borylation
Mozingo reduction
Noyori asymmetric hydrogenation
Omega oxidation
Oppenauer oxidation
Oxygen rebound mechanism
Ozonolysis
Parikh–Doering oxidation
Pinnick oxidation
Prévost reaction
Reduction of nitro compounds
Reductive amination
Riley oxidation
Rosenmund reduction
Rubottom oxidation
Sabatier reaction
Sarett oxidation
Selenoxide elimination
Shapiro reaction
Sharpless asymmetric dihydroxylation
Epoxidation of allylic alcohols
Sharpless epoxidation
Sharpless oxyamination
Stahl oxidation
Staudinger reaction
Stephen aldehyde synthesis
Swern oxidation
Transfer hydrogenation
Wacker process
Wharton reaction
Whiting reaction
Wohl–Aue reaction
Wolff–Kishner reduction
Wolffenstein–Böters reaction
Zinin reaction Rearrangement
1,2-rearrangement
1,2-Wittig rearrangement
2,3-sigmatropic rearrangement
2,3-Wittig rearrangement
Achmatowicz reaction
Alkyne zipper reaction
Allen–Millar–Trippett rearrangement
Allylic rearrangement
Alpha-ketol rearrangement
Amadori rearrangement
Arndt–Eistert reaction
Aza-Cope rearrangement
Baker–Venkataraman rearrangement
Bamberger rearrangement
Banert cascade
Beckmann rearrangement
Benzilic acid rearrangement
Bergman cyclization
Bergmann degradation
Boekelheide reaction
Brook rearrangement
Buchner ring expansion
Carroll rearrangement
Chan rearrangement
Claisen rearrangement
Cope rearrangement
Corey–Fuchs reaction
Cornforth rearrangement
Criegee rearrangement
Curtius rearrangement
Demjanov rearrangement
Di-π-methane rearrangement
Dimroth rearrangement
Divinylcyclopropane-cycloheptadiene rearrangement
Dowd–Beckwith ring-expansion reaction
Electrocyclic reaction
Ene reaction
Enyne metathesis
Favorskii reaction
Favorskii rearrangement
Ferrier carbocyclization
Ferrier rearrangement
Fischer–Hepp rearrangement
Fries rearrangement
Fritsch–Buttenberg–Wiechell rearrangement
Gabriel–Colman rearrangement
Group transfer reaction
Halogen dance rearrangement
Hayashi rearrangement
Hofmann rearrangement
Hofmann–Martius rearrangement
Ireland–Claisen rearrangement
Jacobsen rearrangement
Kornblum–DeLaMare rearrangement
Kowalski ester homologation
Lobry de Bruyn–Van Ekenstein transformation
Lossen rearrangement
McFadyen–Stevens reaction
McLafferty rearrangement
Meyer–Schuster rearrangement
Mislow–Evans rearrangement
Mumm rearrangement
Myers allene synthesis
Nazarov cyclization reaction
Neber rearrangement
Newman–Kwart rearrangement
Overman rearrangement
Oxy-Cope rearrangement
Pericyclic reaction
Piancatelli rearrangement
Pinacol rearrangement
Pummerer rearrangement
Ramberg–Bäcklund reaction
Ring expansion and contraction
Ring-closing metathesis
Rupe reaction
Schmidt reaction
Semipinacol rearrangement
Seyferth–Gilbert homologation
Sigmatropic reaction
Skattebøl rearrangement
Smiles rearrangement
Sommelet–Hauser rearrangement
Stevens rearrangement
Stieglitz rearrangement
Thermal rearrangement of aromatic hydrocarbons
Tiffeneau–Demjanov rearrangement
Vinylcyclopropane rearrangement
Wagner–Meerwein rearrangement
Wallach rearrangement
Weerman degradation
Westphalen–Lettré rearrangement
Willgerodt rearrangement
Wolff rearrangement Ring forming
1,3-Dipolar cycloaddition
Annulation
Azide-alkyne Huisgen cycloaddition
Baeyer–Emmerling indole synthesis
Bartoli indole synthesis
Bergman cyclization
Biginelli reaction
Bischler–Möhlau indole synthesis
Bischler–Napieralski reaction
Blum–Ittah aziridine synthesis
Bobbitt reaction
Bohlmann–Rahtz pyridine synthesis
Borsche–Drechsel cyclization
Bucherer carbazole synthesis
Bucherer–Bergs reaction
Cadogan–Sundberg indole synthesis
Camps quinoline synthesis
Chichibabin pyridine synthesis
Cook–Heilbron thiazole synthesis
Cycloaddition
Darzens reaction
Davis–Beirut reaction
De Kimpe aziridine synthesis
Debus–Radziszewski imidazole synthesis
Dieckmann condensation
Diels–Alder reaction
Feist–Benary synthesis
Ferrario–Ackermann reaction
Fiesselmann thiophene synthesis
Fischer indole synthesis
Fischer oxazole synthesis
Friedländer synthesis
Gewald reaction
Graham reaction
Hantzsch pyridine synthesis
Hegedus indole synthesis
Hemetsberger indole synthesis
Hofmann–Löffler reaction
Hurd–Mori 1,2,3-thiadiazole synthesis
Iodolactonization
Isay reaction
Jacobsen epoxidation
Johnson–Corey–Chaykovsky reaction
Knorr pyrrole synthesis
Knorr quinoline synthesis
Kröhnke pyridine synthesis
Kulinkovich reaction
Larock indole synthesis
Madelung synthesis
Nazarov cyclization reaction
Nenitzescu indole synthesis
Niementowski quinazoline synthesis
Niementowski quinoline synthesis
Paal–Knorr synthesis
Paternò–Büchi reaction
Pechmann condensation
Petrenko-Kritschenko piperidone synthesis
Pictet–Spengler reaction
Pomeranz–Fritsch reaction
Prilezhaev reaction
Pschorr cyclization
Reissert indole synthesis
Ring-closing metathesis
Robinson annulation
Sharpless epoxidation
Simmons–Smith reaction
Skraup reaction
Urech hydantoin synthesis
Van Leusen reaction
Wenker synthesis Cycloaddition
1,3-Dipolar cycloaddition
4+4 Photocycloaddition
(4+3) cycloaddition
6+4 Cycloaddition
Alkyne trimerisation
Aza-Diels–Alder reaction
Azide-alkyne Huisgen cycloaddition
Bradsher cycloaddition
Cheletropic reaction
Conia-ene reaction
Cyclopropanation
Diazoalkane 1,3-dipolar cycloaddition
Diels–Alder reaction
Enone–alkene cycloadditions
Hexadehydro Diels–Alder reaction
Intramolecular Diels–Alder cycloaddition
Inverse electron-demand Diels–Alder reaction
Ketene cycloaddition McCormack reaction
Metal-centered cycloaddition reactions
Nitrone-olefin (3+2) cycloaddition
Oxo-Diels–Alder reaction
Ozonolysis
Pauson–Khand reaction
Povarov reaction
Prato reaction
Retro-Diels–Alder reaction
Staudinger synthesis
Trimethylenemethane cycloaddition
Vinylcyclopropane (5+2) cycloaddition
Wagner-Jauregg reaction Heterocycle forming reactions
Algar–Flynn–Oyamada reaction
Allan–Robinson reaction
Auwers synthesis
Bamberger triazine synthesis
Banert cascade
Barton–Zard reaction
Bernthsen acridine synthesis
Bischler–Napieralski reaction
Bobbitt reaction
Boger pyridine synthesis
Borsche–Drechsel cyclization
Bucherer carbazole synthesis
Bucherer–Bergs reaction
Chichibabin pyridine synthesis
Cook–Heilbron thiazole synthesis
Diazoalkane 1,3-dipolar cycloaddition
Einhorn–Brunner reaction
Erlenmeyer–Plöchl azlactone and amino-acid synthesis
Feist–Benary synthesis
Fischer oxazole synthesis
Gabriel–Colman rearrangement
Gewald reaction
Hantzsch ester
Hantzsch pyridine synthesis
Herz reaction
Knorr pyrrole synthesis
Kröhnke pyridine synthesis
Lectka enantioselective beta-lactam synthesis
Lehmstedt–Tanasescu reaction
Niementowski quinazoline synthesis
Nitrone-olefin (3+2) cycloaddition
Paal–Knorr synthesis
Pellizzari reaction
Pictet–Spengler reaction
Pomeranz–Fritsch reaction
Prilezhaev reaction
Robinson–Gabriel synthesis
Stollé synthesis
Urech hydantoin synthesis
Wenker synthesis
Wohl–Aue reaction
Authority control databases
International National Other