Dichlorine trioxide
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
dichlorine trioxide
| |||
| Other names
chlorine trioxide
chlorine chlorate chlorine(I,V) oxide | |||
| Identifiers | |||
CAS Number
|
|||
3D model (JSmol)
|
|||
| ChemSpider | |||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
Cl2O3 | ||
| Molar mass | 118.903 g/mol | ||
| Appearance | dark brown solid | ||
| Melting point | explodes below 0 °C | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
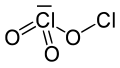
Dichlorine trioxide, Cl2O3, is a chlorine oxide. It is a dark brown solid discovered in 1967 which is explosive even below 0 °C.[2] It is formed by the low-temperature photolysis of ClO2 and is formed along with Cl2O6, Cl2 and O2. Its structure is believed to be OCl−ClO2 with possible isomers such as Cl−O−ClO2.[3] The isomer having a structure of OCl–O–ClO would be the theoretical anhydride of chlorous acid.
References
- ^ Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). Boca Raton, Florida: CRC Press. pp. 4–51. ISBN 0-8493-0594-2.
- ^ N. N. Greenwood and A. Earnshaw (1997). Chemistry of the Elements. Butterworth-Heinemann. ISBN 978-0750633659.
- ^ Egon Wiberg, Arnold Frederick Holleman (2001) Inorganic Chemistry, Elsevier ISBN 0-12-352651-5