Gold(III) oxide
 | |
| Names | |
|---|---|
| IUPAC name
Gold(III) oxide
| |
| Other names
Gold trioxide, Gold sesquioxide, Auric oxide
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.013.748 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Au2O3 |
| Molar mass | 441.93 |
| Appearance | red-brown solid |
| Density | 11.34 g/cm3 at 20 °C[1] |
| Melting point | 298 °C (568 °F; 571 K)[2] |
Solubility in water
|
insoluble in water, soluble in hydrochloric and nitric acid |
| Structure | |
Crystal structure
|
Orthorhombic, oF40 |
Space group
|
= Fdd2, No. 43[1] |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H315, H319 |
Precautionary statements
|
P264, P264+P265, P280, P302+P352, P305+P351+P338, P321, P332+P317, P337+P317, P362+P364 |
| NFPA 704 (fire diamond) | 
1
0
0 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Gold(III) oxide (Au2O3) is an inorganic compound of gold and oxygen with the formula Au2O3. It is a red-brown solid that decomposes at 298 °C.[3]
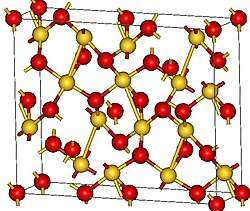
According to X-ray crystallography, Au2O3 features square planar gold centers with both 2- and 3-coordinated oxides. The four Au-O bond distances range from 193 to 207 picometers.[1]
The crystals can be prepared by heating amorphous hydrated gold(III) oxide with perchloric acid and an alkali metal perchlorate in a sealed quartz tube at a temperature of around 250 °C and a pressure of around 30 MPa.[4]
References
- ^ a b c Jones, P. G.; Rumpel, H.; Schwarzmann, E.; Sheldrick, G. M.; Paulus, H. (1979). "Gold(III) oxide". Acta Crystallographica Section B. 35 (6): 1435. doi:10.1107/S0567740879006622.
- ^ Kawamoto, Daisuke; Ando, Hiroaki; Ohashi, Hironori; Kobayashi, Yasuhiro; Honma, Tetsuo; Ishida, Tamao; Tokunaga, Makoto; Okaue, Yoshihiro; Utsunomiya, Satoshi; Yokoyama, Takushi (2016-11-15). "Structure of a Gold(III) Hydroxide and Determination of Its Solubility". Bulletin of the Chemical Society of Japan. 89 (11). The Chemical Society of Japan: 1385–1390. doi:10.1246/bcsj.20160228. ISSN 0009-2673.
- ^ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.
- ^ Jones, Peter G.; Rumpel, Horst; Sheldrick, George M.; Schwarzmann, Einhard (1980). "Gold(III) oxide and oxychloride" (open access). Gold Bulletin. 13 (2): 56. doi:10.1007/BF03215453.
External links
 Media related to Gold(III) oxide at Wikimedia Commons
Media related to Gold(III) oxide at Wikimedia Commons