Stannane
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Stannane
| |||
| Other names
tin tetrahydride
tin hydride tin(IV) hydride | |||
| Identifiers | |||
CAS Number
|
|||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
SnH4 | ||
| Molar mass | 122.71 g/mol | ||
| Appearance | colourless gas | ||
| Density | 5.4 g/L, gas | ||
| Melting point | −146 °C (−231 °F; 127 K) | ||
| Boiling point | −52 °C (−62 °F; 221 K) | ||
| Structure | |||
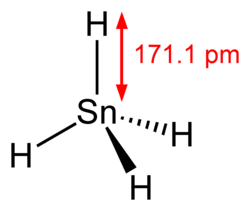
Molecular shape
|
Tetrahedral | ||
Dipole moment
|
0 D | ||
| Thermochemistry | |||
Heat capacity (C)
|
1.262 kJ/(kg·K) | ||
Std enthalpy of
formation (ΔfH⦵298) |
162.8 kJ/mol | ||
Enthalpy of vaporization (ΔfHvap)
|
19.049 kJ/mol | ||
| Related compounds | |||
Related organotins
|
tributylstannane (Bu3SnH) | ||
Related compounds
|
Methane Silane Germane Plumbane | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
Stannane /ˈstæneɪn/ or tin hydride is an inorganic compound with the chemical formula SnH4. It is a colourless gas that ignites on contact with air. SnH4 is a heavy analogue of methane but much less robust. In contrast to this rarely used species, stannane also refers to a large collection of organotin compounds.
Preparation and reactions
Stannane can be prepared by the reaction of SnCl4 and lithium aluminium hydride according to the following idealized equation:[1]
- SnCl4 + Li[AlH4] → SnH4 + LiCl + AlCl3
Stannane decomposes slowly at room temperature to give metallic tin and hydrogen.[1]
- SnH4 → Sn + 2H2
Organotin hydrides are more stable, e.g. triphenyltin hydride, also known as triphenylstannane.
Occurrence in industry
The volatility of stannane is potentially relevant to production of microchips.[2]
See also
- Organotin
References
- ^ a b Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. doi:10.1016/C2009-0-30414-6. ISBN 978-0-08-037941-8.
- ^ Qerimi, Dren; Herschberg, Andrew C.; Panici, Gianluca; Hays, Parker; Pohlman, Tyler; Ruzic, David N. (2022). "Tin removal by an annular surface wave plasma antenna in an extreme ultraviolet lithography source". Journal of Applied Physics. 132 (11) 113302. doi:10.1063/5.0094375. OSTI 1887630.

