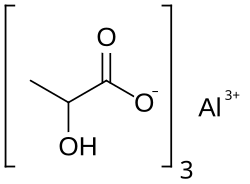
Aluminium lactate
 | |
| Names | |
|---|---|
| Other names
Aluminium trilactate, tris(2-hydroxypropanoato)aluminium
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.038.776 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
C9H15AlO9 |
| Molar mass | 294.192 g·mol−1 |
| Appearance | White powder |
| Melting point | 300 °C (572 °F; 573 K) |
Solubility in water
|
Soluble |
| Hazards | |
| GHS labelling: | |
Pictograms
|

|
Signal word
|
Warning |
Hazard statements
|
H315, H319, H335 |
Precautionary statements
|
P302, P305, P338, P351, P352 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Aluminium lactate is a chemical compound, a salt of aluminium and lactic acid with the formula Al(C3H5O3)3.[1][2]
Synthesis
Aluminium lactate is obtained by precipitating a solution of the barium salt by aluminium sulfate.[3]
Physical properties
Aluminium lactate appears as a white powder which is soluble in water.
Use
Aluminium lactate is used as a mordant.[4]
It is suitable for use in the cosmetic industry as a keratolytic agent[5] or antiperspirant,[6] and in the oral industry.[7][8]
Aluminium lactate is also used as a precursor for sol–gel synthesis of alumina-based glasses.[9]
References
- ^ Vargel, Christian (12 May 2020). Corrosion of Aluminium. Elsevier. p. 748. ISBN 978-0-08-099927-2. Retrieved 24 January 2022.
- ^ "Aluminum L-lactate". American Elements. Retrieved 24 January 2022.
- ^ Proceedings of the American Pharmaceutical Association at the Annual Meeting. American Pharmaceutical Association. 1887. p. 291. Retrieved 24 January 2022.
- ^ "Aluminium Lactate - mordant for natural dyeing plant (cellulose) fibres". DT Craft and Design. 14 August 2021. Retrieved 24 January 2022.
- ^ Hunt, Laura; Tankeu, Raissa; Thilk, Alexia; Coppenrath, Valerie (2014). "Ammonium Lactate–Containing Moisturizers: A Systematic Review". U.S. Pharmacist. 39 (11): 46–49. Retrieved 24 January 2022.
- ^ Karsai, Syrus; Weiß, Christel; Lütgerath, Constantin; Ott, Isabel; Faulhaber, Jörg (July 2021). "Comparison of novel aluminium lactate versus aluminium chloride-based antiperspirant in excessive axillary perspiration: First prospective cohort study". Dermatologic Therapy. 34 (4) e15020. doi:10.1111/dth.15020. PMID 34085372.
- ^ "Aluminium Lactate by DPL-US - Personal Care & Cosmetics". ulprospector.com. Retrieved 24 January 2022.
- ^ Lussi, Adrian (1 January 2006). Dental Erosion: From Diagnosis to Therapy. Karger Medical and Scientific Publishers. p. 182. ISBN 978-3-8055-8097-7. Retrieved 24 January 2022.
- ^ Zhang, Long; de Araujo, Carla C.; Eckert, Hellmut (May 2007). "Aluminum lactate – An attractive precursor for sol–gel synthesis of alumina-based glasses". Journal of Non-Crystalline Solids. 353 (13–15): 1255–1260. Bibcode:2007JNCS..353.1255Z. doi:10.1016/j.jnoncrysol.2006.10.065.