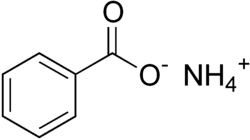
Ammonium benzoate
 | |||
| |||
| Identifiers | |||
|---|---|---|---|
CAS Number
|
|||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.015.881 | ||
| EC Number |
| ||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
InChI
| |||
SMILES
| |||
| Properties | |||
Chemical formula
|
C7H9NO2 | ||
| Molar mass | 139.15 g/mol | ||
| Appearance | White solid | ||
| Density | 1.26 g/cm3 | ||
| Melting point | 198 °C (388 °F; 471 K) | ||
Solubility in water
|
21.3 g/100 mL (20 °C) 83 g/100 mL (100 °C) | ||
| Solubility | soluble in methanol insoluble in diethyl ether | ||
| Hazards | |||
| GHS labelling: | |||
Pictograms
|

| ||
Signal word
|
Warning | ||
Hazard statements
|
H302, H315, H319, H335 | ||
Precautionary statements
|
P261, P264, P270, P271, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362, P403+P233, P405, P501 | ||
| NFPA 704 (fire diamond) | 
2
1
2 | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
825 mg/kg, oral (rat) | ||
| Safety data sheet (SDS) | Fisher Scientific | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |||
Ammonium benzoate, a white powder-like substance, is the ammonium salt of benzoic acid.[1] This compound is prepared by the reaction of benzoic acid and ammonia.
Uses
Ammonium benzoate is used as a food preservative, due to its antibacterial and antifungal properties.[2][3]
Reactions
Ammonium benzoate can be dehydrated to form benzamide:
C7H9NO2 → C7H7NO + H2O
References
- ^ Yang, Wei-Wei; Di, You-Ying; Kong, Yu-Xia; Guo, Xiao-Yang; Tan, Zhi-Cheng (2010). "Synthesis, characterization, and thermodynamic study of ammonium benzoate C7H5O2NH4(s)". Thermochimica Acta. 502 (1–2): 14–19. doi:10.1016/j.tca.2010.01.021.
- ^ "Ammonium Benzoate: Essential Properties and Applications for Manufacturers". www.nbinno.com. Retrieved 2026-01-12.
- ^ "21 CFR 175.105 - Adhesives". www.ecfr.gov. Retrieved 2026-01-12.