Diammonium tetrachloroplatinate
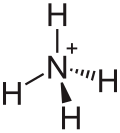 2 2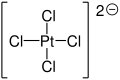
| |
| Names | |
|---|---|
| IUPAC name
diazanium;tetrachloroplatinum(2-)
| |
| Other names
Ammonium tetrachloroplatinate(II)
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.034.076 |
| EC Number |
|
Gmelin Reference
|
79515 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
Cl4H8N2Pt |
| Molar mass | 372.96 g·mol−1 |
| Appearance | red crystals |
| Density | 2,94 g/cm3 |
| Melting point | 140 °C |
Solubility in water
|
soluble |
| Hazards | |
| GHS labelling:[1] | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H301, H315, H317, H318, H334 |
Precautionary statements
|
P280, P301, P302, P305, P310, P330, P338, P351, P352 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Diammonium tetrachloroplatinate is a chemical compound with the chemical formula (NH4)2[PtCl4].[2][3]
Synthesis
Diammonium tetrachloroplatinate can be synthesised by the reduction of ammonium hexachloroplatinate(IV) with ammonium oxalate:
- (NH4)2[PtCl6] + (NH4)2C2O4 → (NH4)2[PtCl4] + 2NH4Cl + 2CO2
Physical properties
Diammonium tetrachloroplatinate forms odorless red crystals,[4] soluble in water, insoluble in ethanol.[1][5]
The crystals have a cubic structure with the space group P4/mmm (space group number 123).
Chemical properties
Diammonium tetrachloroplatinate decomposes if heated:
- (NH4)2[PtCl4] → PtCl2 + 2NH4Cl
Uses
Diammonium tetrachloroplatinate was used in photography.[6][7] Also used in spectral analysis standard and in the preparation of platinum sponge and platinum catalytic agent.[4]
References
- ^ a b "Ammoniumtetrachloroplatinat" (in German). gestis.dguv.de. Retrieved 11 March 2025.
- ^ "Ammonium tetrachloroplatinate(II)". Sigma Aldrich. Retrieved 11 March 2025.
- ^ "WebElements Periodic Table » Platinum » diammonium tetrachloroplatinate". winter.group.shef.ac.uk. Retrieved 11 March 2025.
- ^ a b "Ammonium tetrachloroplatinate(II), 99.9% (metals basis), Pt 51% min, Thermo Scientific Chemicals | Fisher Scientific". Fisher Scientific. Retrieved 11 March 2025.
- ^ Perry, Dale L. (19 April 2016). Handbook of Inorganic Compounds. CRC Press. p. 34. ISBN 978-1-4398-1462-8. Retrieved 12 March 2025.
- ^ Malde, Pradip; Ware, Mike (30 December 2020). Platinotype: Making Photographs in Platinum and Palladium with the Contemporary Printing-out Process. Routledge. p. 63. ISBN 978-1-000-28116-3. Retrieved 12 March 2025.
- ^ Kanerva, Lasse (2000). Handbook of Occupational Dermatology. Springer Science & Business Media. p. 1194. ISBN 978-3-540-64046-2. Retrieved 11 March 2025.