Cadmium sulfate
.jpg) | |
| Names | |
|---|---|
| IUPAC name
Cadmium(II) sulfate
| |
| Other names
Sulfuric acid, cadmium salt (1:1),
| |
| Identifiers | |
CAS Number
|
|
3D model (JSmol)
|
|
| ChEBI |
|
| ChemSpider | |
| ECHA InfoCard | 100.030.288 |
| EC Number |
|
Gmelin Reference
|
8295 |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
| UN number | 2570 |
CompTox Dashboard (EPA)
|
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula
|
|
| Molar mass |
|
| Appearance | White hygroscopic solid |
| Odor | odorless |
| Density |
|
| Melting point |
|
| Boiling point | (decomposes to basic sulfate and then oxide) |
Solubility in water
|
|
| Solubility in methanol | slightly soluble |
| Solubility in ethyl acetate | slightly soluble |
Magnetic susceptibility (χ)
|
−59.2×10−6 cm3/mol |
Refractive index (nD)
|
1.565 |
| Viscosity |
|
| Structure | |
Crystal structure
|
|
| Thermochemistry | |
Std molar
entropy (S⦵298) |
123 J⋅mol−1·K-1[2] |
Std enthalpy of
formation (ΔfH⦵298) |
−935 kJ⋅mol−1[2] |
| Hazards[4] | |
| GHS labelling: | |
Pictograms
|
  
|
Signal word
|
Danger |
Hazard statements
|
H301, H330, H340, H350, H360, H372, H410 |
Precautionary statements
|
P201, P202, P260, P264, P270, P271, P273, P280, P284, P301+P310+P330, P304+P340+P310, P308+P313, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 
3
1
1 |
Threshold limit value (TLV)
|
|
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
107 mg/kg (oral, rat, analgous compound) |
LC50 (median concentration)
|
0.75 mg/L (goldfish, analgous compound) |
| NIOSH (US health exposure limits):[5] | |
PEL (Permissible)
|
TWA 0.005 mg/m3 (as Cd) |
REL (Recommended)
|
TWA 0.005 mg/m3 (as Cd) |
IDLH (Immediate danger)
|
9 mg/m3 (as Cd) |
| Safety data sheet (SDS) | |
| Related compounds | |
Other anions
|
|
Other cations
|
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
Infobox references
| |
Cadmium sulfate is the name of a series of related inorganic compounds with the formula CdSO4·xH2O. The most common form is the monohydrate CdSO4·H2O, but two other forms are known: the octahydrate (3CdSO4·8H2O) and the anhydrous salt (CdSO4). All salts are colourless and highly soluble in water.
Preparation
Cadmium sulfate hydrate can be prepared by the reaction of cadmium metal or its oxide or hydroxide with dilute sulfuric acid:
- CdO + H2SO4 → CdSO4 + H2O
- Cd + H2SO4 → CdSO4 + H2
The anhydrous material can be prepared using sodium persulfate:
- Cd + Na2S2O8 → CdSO4 + Na2SO4
Applications
Cadmium sulfate is used widely for the electroplating of cadmium in electronic circuits. It is also a precursor to cadmium-based pigment such as cadmium sulfide. It is also used for electrolyte in a Weston standard cell as well as a pigment in fluorescent screens.
Structure
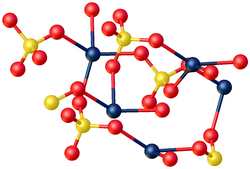
X-ray crystallography shows that CdSO4·H2O is a typical coordination polymer. Each Cd2+ center has octahedral coordination geometry, being surrounded by four oxygen centers provided by four sulfate ligands and two oxygen centers from the bridging water ligands.[7]
Occurrence
Cadmium sulfates occur as the following rare minerals drobecite (CdSO4·4H2O), voudourisite (monohydrate), and lazaridisite (the octahydrate).
Safety
Cadmium sulfate (along with cadmium and its compounds) are classified as group 1 (human carcinogens) by IARC and have been identified as causing lung and prostate cancer as well as mutagenic effects in humans.[8]
References
- ^ Lide, David R., ed. (2006). CRC Handbook of Chemistry and Physics (87th ed.). Boca Raton, Florida: CRC Press. ISBN 0-8493-0487-3.
- ^ a b Zumdahl, Steven S. (2009). Chemical Principles 6th Ed. Houghton Mifflin Company. p. A21. ISBN 978-0-618-94690-7.
- ^ a b "Safety Data Sheet - Cadmium Sulfate". fishersci.com. ThermoFisher Scientific. 28 December 2021. p. 3. Retrieved 21 October 2025.
- ^ a b "Safety Data Sheet - Cadmium Sulfate". sigmaaldrich.com. Sigma-Aldrich. 20 May 2025. Retrieved 21 October 2025.
- ^ "NIOSH Pocket Guide to Chemical Hazards".
- ^ Aurivillius, Karin; Stålhandske, Claes (1980). "A Reinvestigation of the Crystal Structures of HgSO4 and CdSO4". Zeitschrift für Kristallographie - Crystalline Materials. 153 (1–2): 121–129. Bibcode:1980ZK....153..121A. doi:10.1524/zkri.1980.0011.
- ^ Theppitak, C.; Chainok, K. (2015). "Crystal Structure of CdSO4(H2O): A Redetermination". Acta Crystallographica Section E. 71 (10): i8–pi9. doi:10.1107/S2056989015016904. PMC 4647421. PMID 26594423.
- ^ "Cadmium". Beryllium, Cadmium, Mercury, and Exposures in the Glass Manufacturing Industry (PDF). Lyon, France: World Health Organization - International Agency for Research on Cancer. February 1993. p. 210. ISBN 92-832-1258-4. ISSN 0250-9555. Retrieved 21 October 2025.